Assessment of Dried Pineapple By-products as a Bioactive Plant Material for Enhancing Dairy Productivity in Holstein Cows
This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Agro-industrial by-products represent an important opportunity for sustainable livestock production. Pineapple (Ananas comosus) residues are rich in bromelain, polyphenols, and dietary fiber and are known for their antioxidant and anti-inflammatory activities. However, few studies have evaluated the effects of dried pineapple by-product powder (DPP) on dairy productivity and metabolic function.
A DPP feeding trial was conducted with 20 mid-lactation Holstein cows (90 ± 15 days old in milk; 620 ± 30 kg body weight). Cows were randomly assigned to two groups: a control group fed a basal total mixed ration (TMR) and a treatment, fed a TMR supplemented with 1% DPP (dry matter basis) for 4 weeks. Milk yield and composition were measured weekly, and blood samples were analyzed at weeks 0 and 8 for serum biochemical parameters including glucose, amylase, and lipase. Cows fed DPP showed a 6.3% increase in daily milk yield (p < 0.05) without significant changes in milk fat, protein, or lactose concentrations. The somatic cell count (SCC) decreased by 23% (p < 0.05), indicating improved udder health. Blood glucose concentrations decreased (p < 0.05), suggesting improved energy utilization for milk synthesis, without changes in amylase or lipase activity. Liver (aspartate aminotransferase and alanine aminotransferase) and kidney (creatinine) function remained within normal physiological limits. Based on the in vivo findings, the antioxidant, and anti-inflammatory activities of DPP were evaluated in vitro. The ABTS radical scavenging assay demonstrated strong antioxidant capacity (IC50≈ 220 µg/mL), and the nitric oxide (NO) inhibition assay showed significant, dose-dependent suppression of NO production in LPS-stimulated RAW 264.7 macrophages (p < 0.01), confirming antioxidant and anti-inflammatory effects.
DPP improved in vivo dairy performance by enhancing milk yield, reducing SCC, supporting efficient energy metabolism without adverse effects on liver or kidney function, and exhibiting potent in vitro antioxidant and anti-inflammatory properties. These findings highlight the potential of DPP as a sustainable and functional feed additive for dairy cows.
Keywords:
Lactating Dairy Cow, Dried Pineapple By-product Powder, Milk Productivity, Antioxidant, NO AssayINTRODUCTION
Pineapple (Ananas comosus), a member of the family Bromeliaceae, comprises more than 2,000 species and has long been recognized not only as a nutrient-dense tropical fruit but also as a bioactive medicinal crop with diverse therapeutic potentials. Beyond its well-known nutritional value—rich sugars, minerals, dietary fibres, organic acids, and vitamins including carotenoids, ascorbic acid, and flavonoids—the plant contains phytochemicals with documented pharmacological activities, making it relevant in medicinal crop science (Ketnawa et al., 2012). The phytochemical profile of pineapple varies by cultivar and geographic origin, yet consistently demonstrates high functional value. A single fresh pineapple can provide approximately 17% of the daily vitamin C requirement, and is abundant in B-complex vitamins such as pyridoxine, folate, riboflavin, and niacin, contributing to antioxidant defense and metabolic support (Mehraj et al., 2024). Importantly, pineapple and its by-products contain bromelain, a proteolytic enzyme complex widely studied as a natural anti-inflammatory, anti-thrombotic, fibrinolytic, digestive, and anti-cancer agent. Recent studies have reinforced bromelain’s potential roles in modulating immune responses, attenuating oxidative stress, reducing edema, improving gastrointestinal function, and even contributing to metabolic regulation (Ajayi et al., 2022; Habotta et al., 2022; Sukri et al., 2023).
Milk yield is a critical parameter in dairy farming, directly influencing the profitability and sustainability of dairy operations. While genetic selection has led to significant improvements in milk production over recent decades, environmental and nutritional factors remain pivotal in realizing the full genetic potential of dairy cows (Brito et al., 2021). Among these, the use of feed additives and nutritional supplements aimed at enhancing lactation performance has gained increasing attention. Milk yield-enhancing supplements work through a variety of physiological mechanisms. These include improving dry matter intake (DMI), optimizing rumen fermentation, enhancing energy metabolism, and supporting mammary gland function (Kholif et al., 2021). Commonly used supplements include rumen-protected amino acids, niacin, yeast culture, monensin, and various phytogenic compounds. For example, rumen-protected choline has been shown to reduce hepatic lipid accumulation and support liver function during early lactation, leading to improved milk production (Ghaffari et al., 2025). In addition to nutrient-based supplements, additives that modulate stress responses or immune function, such as antioxidants, probiotics, and adaptogens have also demonstrated promising results in supporting lactation under challenging conditions (Kalogerakou and Antoniadou, 2024). Moreover, certain feed additives that improve water intake or mitigate heat stress have indirect but significant effects on milk yield.
Given the increasing demand for sustainable and efficient dairy production, understanding the role of supplements in optimizing milk yield is of both economic and scientific importance. Pineapple meal is a naturally occurring amino acid derivative involved in mitochondrial function and antioxidative defense. In livestock, pineapple meal has shown promise in promoting growth and enhancing physiological functions. This study investigates the potential benefits of Pineapple meal supplementation in dairy cattle, with particular focus on milk yield, somatic cell count (SCC), and changes in blood profiles (Dhiman et al., 2025).
Taken together, these findings suggest that pineapple extract either in crude or purified form holds promise as a functional feed additive to improve digestive efficiency, lactational performance, and overall health in dairy cattle. However, direct evidence regarding the dietary application of concentrated pineapple extract in dairy cows remains limited. Therefore, the present study aims to investigate its potential effects on milk yield, composition, and animal health.
MATERIALS AND METHODS
1. Dried pineapple by-product powder
The dried DPP was produced from peel, core, and pulp residues collected during the pineapple juice manufacturing process. These by-products were heat-dried and then finely ground into powder form. The DPP was determined to be 13.58 CDU/mg.
2. Animals
All experimental procedures were conducted in accordance with the guidelines of the Institutional Animal Care and Use Committee (IACUC) of Chonnam National University (Approval No.: CNU IACUC-YB-2025-32). A total of twenty Holstein dairy cows with an average age of 52 months (ranging from the 1st to 6th parity) were used in this study. The experiment was conducted at a commercial dairy farm located in Sangju, Gyeongsangbuk-do, Republic of Korea, where the animals were housed in a sawdust-bedded free-stall barn.
The animals were fed a total mixed ration (TMR) once daily at 17:00, with an allowance of 30 kg per cow per day. Feed refusals were collected the following day at 16:00 to determine the average feed intake per group. During the experimental period, the dry matter intake was recorded as 22.38 ± 1.39 kg per cow per day.
To evaluate the effects of DPP supplementation on milk production performance and nutrient metabolism in lactating Holstein cows, milk yield, milk composition, and blood biochemical parameters were analyzed in control and treatment groups.
Experimental diets were formulated according to the National Research Council (NRC) (2001) feed library nutrient requirements for lactating dairy cows, based on recommended dry matter intake and nutrient composition. The proximate composition, neutral detergent fiber, and acid detergent fiber contents of the feed ingredients were determined by chemical analysis, while any missing nutrient data were supplemented using values listed in the NRC feed library.
In the treatment group, 1.00% of soybean hulls in the control diet was replaced with DPP. The ingredient composition and chemical analysis of the experimental diets are shown in Table 1.
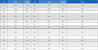
TMR formula and chemical composition of control and dried pineapple by-product powder (DPP) diet in dairy lactating cows.
The experiment lasted for a total of four weeks, consisting of two weeks of control diet adaptation followed by two weeks of treatment diet adaptation. For data analysis, samples and measurements were collected from both groups on day 14 of each feeding period.
3. Proximate composition analysis
Feed ingredient samples were oven-dried in a forced-air drying oven and ground using a Wiley mill (1 mm screen; Arthur A. Thomas Co., Philadelphia, PA, USA). Moisture, crude protein (CP), ether extract (EE), and ash contents were analyzed according to the AOAC (1990) methods. The neutral detergent fiber (NDF) was determined following the procedure of Van Soest et al. (1991), with the addition of 100 µL of heat-stable α-amylase (A3306; Sigma Chemical Co., St. Louis, MO, USA) per 0.5 g sample during analysis.
4. Measurement of milk yield and sampling
Milk yield was automatically recorded using a 2 × 10 side-by-side milking system (Westfalia Co., Germany) at morning (05:00 h) and evening (16:00 h) milkings throughout the experimental period. Milk samples (20 mL each) were collected from both morning and evening milkings on day 14 of each treatment using an automatic sampler attached to the milking system. Broad Spectrum Microtabs® II (Advanced Instruments, Inc.) were added as preservatives, and the samples were stored at 4℃ until analysis.
Milk composition was analyzed using a Milko-Scan FT 6000 (Foss Electric Co., Hillerød, Denmark) based on the near-infrared spectrophotometric method.
5. Measurement of hematological parameters and sampling
Blood samples were collected from the jugular vein at both time points. Blood was collected into 4 mL tubes (Venosafe, Terumo, Guyancourt, France) from the vein and carefully mixed by inversion. Tubes were immediately refrigerated at ~4℃ until hematologic analysis at the Laboratoire Central de Biologie Médicale (Central Laboratory of Clinical Pathology) of ENVT within 24 h of blood collection.
Biochemical parameters, including glucose, amylase, lipase, alanine aminotransferase (ALT), blood urea nitrogen (BUN), and creatinine, were analyzed using an automated biochemical analyzer (VB1, skyla corporation, Tawian) following the manufacturer’s protocols using commercial kits (Labtest commercial kits, Lagoa Santa, MG, Brazil).
6. ABTS Radical Scavenging Activity
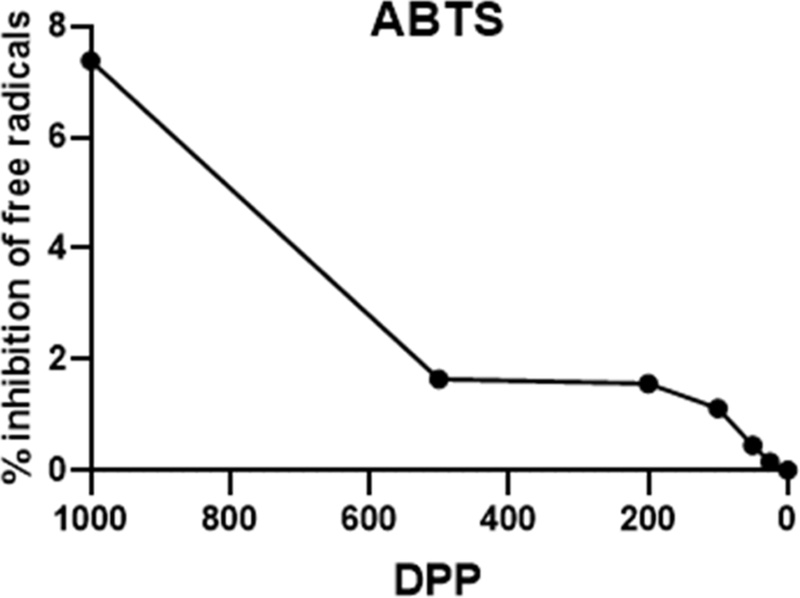
The antioxidant capacity of the DPP extract was evaluated using the 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) assay (n = 5 per group) following the method of (Seo et al., 2025). The extract (0, 25, 50, 100, 200, 500, and 1000 μg/mL) was mixed with ABTS working solution in methanol and incubated at 37℃ for 10 min. Absorbance was measured at 734 nm, and the radical scavenging activity was calculated as:
Scavenging rate (%) = [(Abfree − Absample)/Abfree] × 100
The DPP extract exhibited a dose-dependent increase in ABTS radical scavenging activity, showing strong antioxidant potential with an estimated IC₅₀ of approximately 220 µg/mL.
7. Measurement of nitric oxide (NO), and interleukin-6 (IL-6)
The murine macrophage cell line RAW 264.7 (ATCC, Rockville, MD, USA) was cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; Gibco, Grand Island, NY, USA) containing 5.5% heat-inactivated fetal bovine serum (Gibco), 100 U/mL penicillin, and 100 μg/mL streptomycin. Cells were maintained at 37°C in a humidified incubator with 5% CO2.
For the assay, cells were seeded in 48-well plates at a density of 2.5 × 105 cells/well and allowed to adhere overnight. They were then stimulated with lipopolysaccharide (LPS; 1 μg/mL) in the presence or absence of various concentrations of DPP for 18 h. After incubation, culture supernatants were collected. Nitric oxide (NO) production was measured using the Griess Reagent System (Promega, Madison, WI, USA), and interleukin-6 (IL-6) levels were determined with an IL-6 ELISA kit (BioSource International, Camarillo, CA, USA), according to the manufacturers’ instructions.
8. Statistical analysis
The analysis parameters are presented as mean ± standard error (SE). Statistical significance was identified by unpaired t-test analysis for variance, using GraphPad Prism 8 (GraphPad software, San Diego, CA, USA). Statistical significance was set at p < 0.05.
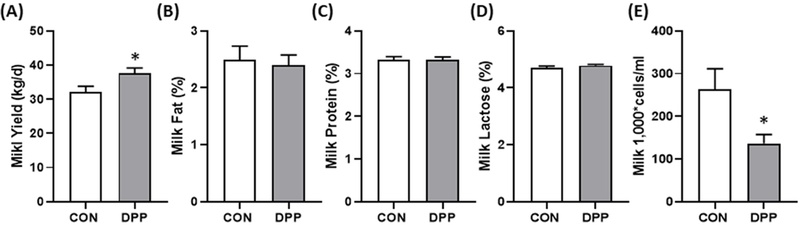
The Changes of milk yield and composition of daily cows fed with DPP.(A) Total milk yield in day, (B) Fat, (C) Protein, and (D) Lactose composition in milk. (E) somatic cell count (SCC) in milk (n = 20 holstein cows/groups). All values are expressed as the mean ± standard error (SE). * p < 0.05 vs. CON group.
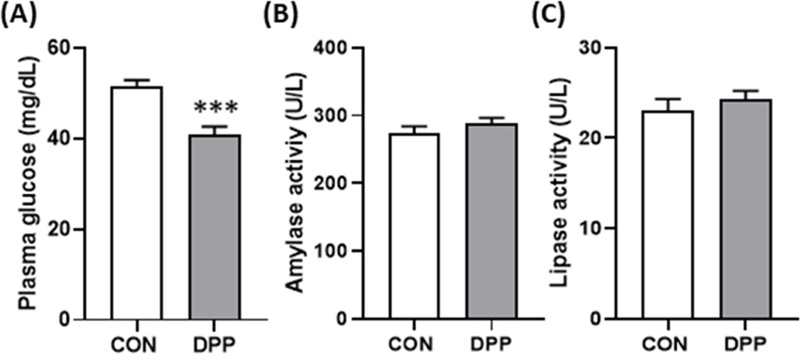
Effects of DPP on biochemical parameters in blood of daily cow.(A) plasma glucose level, (B) amylase, and (C) Lipase activity levels in blood serum (n = 20 holstein cows/groups). All values are expressed as the mean ± standard error (SE). * p < 0.05, and ***p < 0.001 vs. CON group.
RESULTS
1. Milk yield and composition
Feeding trials in twenty mid-lactation Holstein cows demonstrated clear productivity benefits. Cows receiving 1% DPP in the total mixed ration (TMR) showed an average daily milk yield of 37.5 ± 1.62 kg, compared with 32.0 ± 1.81 kg in the control group a 6.3% increase (p < 0.05). Despite this increase in yield, milk fat (2.4 ± 0.18%), protein (3.32 ± 0.07%), and lactose (4.79 ± 0.04%) contents remained unchanged between groups (p > 0.05), suggesting that productivity gains were not accompanied by compositional dilution.
The SCC, an indicator of udder inflammation, was significantly reduced in the treatment group (1.36 × 105 vs. 2.63 × 105 cells/mL; p < 0.05), indicating improved mammary gland health and reduced subclinical mastitis risk.
2. Blood biochemical parameters
Serum biochemical analysis revealed metabolic changes consistent with improved energy balance. Blood glucose levels decreased significantly in the treatment group (40.9 ± 1.73 vs. 51.5 ± 6.26 mg/dL; p < 0.001), suggesting enhanced glucose utilization for lactose synthesis.
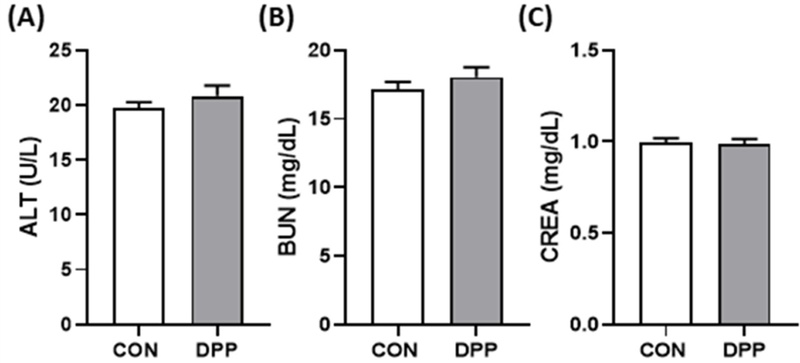
Amylase activity that implying improved digestive or pancreatic enzyme function, and lipase activity remained unchanged (p > 0.05). Indicators of liver and kidney function—ALT (19.7 ± 0.55 vs. 20.8 ± 0.96 U/L), BUN (17.2 ± 0.54 vs. 18.0 ± 0.72 mg/dL), and creatinine (1.00 ± 0.02 vs. 0.99 ± 0.03 mg/dL)—showed no significant differences, remaining within normal physiological ranges for healthy dairy cows.
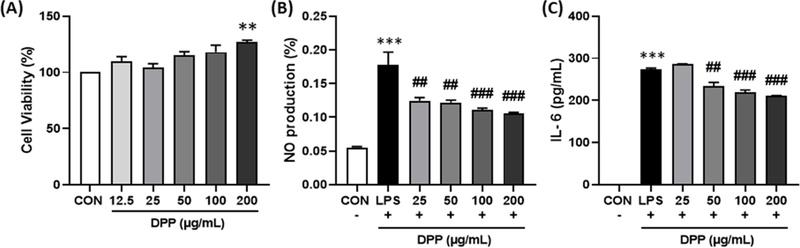
Effects of DPP on cell viability, NO production, and cytokine (IL-6) release in RAW 264.7 cells.The cells were incubated with DPP for 24 h. (A) Cell viability (B) NO production were determined using a Griess reagent assay. (C) IL-6 production in RAW 264.7 cell culture supernatant was determined using an ELISA kit. All values are expressed as the mean ± standard error (SE). * p < 0.05, ** p < 0.01, and *** p < 0.001 vs. CON group. # p < 0.05, ## p < 0.01, and ### p < 0.001 vs. lipopolysaccharides (LPS) group.
3. In vitro antioxidant and anti-inflammatory effects
The antioxidant and anti-inflammatory activities of the DPP were first evaluated using in vitro assays. The ABTS radical scavenging assay revealed a strong antioxidant effect, with scavenging activity increasing in a dose-dependent manner and an IC50 value of approximately 220 µg/mL. In the anti-inflammatory assay using RAW 264.7 macrophages, no cytotoxicity was observed at concentrations up to 200 µg/mL, as confirmed by the CCK-8 assay. Treatment with the extract significantly reduced nitric oxide (NO) and interleukin-6 (IL-6) production in lipopolysaccharide (LPS)-stimulated cells (p < 0.01), demonstrating potent anti-inflammatory activity.
DISCUSSION
The bromelain present in pineapple extract likely contributed to improved protein digestibility and nutrient utilization, thereby supporting increased milk yield and quality (Hawanis et al., 2025; Bruce and Boateng, 2025). The antioxidant and anti-inflammatory properties of pineapple polyphenols and vitamin C improved systemic health and reduced somatic cell count, suggesting potential benefits for mastitis prevention (Hawanis, 2025; Bruce and Boateng, 2025). Moreover, modulation of rumen microbial communities supports the hypothesis that pineapple extract enhances fermentation efficiency. These findings highlight the dual advantage of using DPP: improving dairy cow performance and contributing to sustainable agriculture by valorizing fruit-processing residues (Chen et al., 2022).
Dietary inclusion of DPP improved milk yield and reduced SCC without affecting milk composition, indicating enhanced udder health and productive performance. Similar outcomes have been observed when dairy cows were supplemented with natural antioxidant sources such as grape pomace, or herbal extracts (Santos et al., 2014). The improvement in milk yield may be attributed to enhanced nutrient digestibility and rumen fermentation efficiency. Pineapple residues contain bromelain and soluble carbohydrates that stimulate rumen microbial activity and increase volatile fatty acid production, thereby improving energy supply for milk synthesis (Ketnawa et al., 2011; Gowda et al., 2015; Kyawt et al., 2020).
The significant reduction in SCC suggests anti-inflammatory effects in the mammary gland. High SCC is strongly associated with intramammary infection and oxidative stress in dairy cows (Khan et al., 2023). DPP are rich in phenolic acids, flavonoids, and ascorbic acid, which exert antioxidative and anti-inflammatory effects by scavenging free radicals and suppressing NO production (Polanía et al., 2023). The observed inhibition of NO production in vitro supports this mechanism, consistent with earlier findings that plant polyphenols downregulate iNOS and NF-κB signaling pathways (Sahakyan, 2026).
The antioxidant properties observed in the ABTS assay (IC50 ≈ 220 µg/mL) indicate strong free radical–scavenging potential, similar to other fruit by-products used in ruminant diets (Jang et al., 2020). Antioxidants play a key role in maintaining mammary epithelial integrity and reducing lipid peroxidation, thereby improving milk yield and quality. The concurrent decrease in SCC and stable milk composition further confirm a reduction in subclinical mastitis risk (Somagond et al., 2025).
Serum biochemical parameters remained within normal physiological limits, indicating that DPP supplementation did not adversely affect hepatic or metabolic functions. The decrease in amylase concentration may suggest improved digestive efficiency rather than enzyme suppression. Consistent findings were reported in studies using fruit-derived feed additives, where metabolic enzyme levels remained stable and overall liver health was maintained (Abbasi et al., 2015; Zhu et al., 2025).
Overall, these results indicate that DPP enhance milk yield and udder health primarily through antioxidant and anti-inflammatory mechanisms while maintaining metabolic safety. Their use aligns with the current trend toward sustainable feed resources that improve production efficiency and reduce environmental waste (Anis Mohamad Sukri et al., 2021). Therefore, DPP represent a viable functional and eco-friendly feed additive in dairy nutrition.
In conclusion, DPP supplementation demonstrated antioxidant and anti-inflammatory potential. In lactating cows, supplementation improved milk yield, lowered SCC, and enhanced metabolic efficiency by reducing glucose while increasing amylase activity, with no adverse hepatic or renal effects. These results confirm the safety and functional efficacy of DPP as a sustainable feed additive for improving productivity and udder health in dairy cows.
Acknowledgments
This result was supported by the “Regional Innovation System & Education (RISE)” through the Gwangju RISE Center, funded by the Ministry of Education (MOE) and the Gwangju Metropolitan Government, Republic of Korea(2025-RISE-05-011).
References
-
Abbasi H, Seidavi A, Liu W and Asadpour L. (2015). Investigation on the effect of different levels of dried sweet orange(Citrus sinensis) pulp on performance, carcass characteristics and physiological and biochemical parameters in broiler chicken. Saudi Journal of Biological Sciences. 22:139-146.
[https://doi.org/10.1016/j.sjbs.2014.09.006]

-
Ajayi AM, Coker AI, Oyebanjo OT, Adebanjo IM and Ademowo, OG. (2022). Ananas comosus(L) Merrill(pineapple) fruit peel extract demonstrates antimalarial, anti-nociceptive and anti-inflammatory activities in experimental models. Journal of Ethnopharmacology. 282:114576.
[https://doi.org/10.1016/j.jep.2021.114576]

-
Anis Mohamad Sukri S, Andu Y, Tuan Harith Z, Sarijan S, Naim Firdaus Pauzi M, Seong Wei L, Dawood MAO, and Abdul Kari Z. (2022). Effect of feeding pineapple waste on growth performance, texture quality and flesh colour of nile tilapia(Oreochromis niloticus) fingerlings. Saudi Journal of Biological Sciences. 29:2514-2519.
[https://doi.org/10.1016/j.sjbs.2021.12.027]

- Association of Official Analytical Chemists. (1990) Edition, Association of Official Analytical Chemist, 21th, Washington DC.
-
Assumi S, Jha S and Kaur C. (2018). Valorization of pineapple waste for development of animal feed block. International Journal of Current Microbiology and Applied Sciences. 7:3787-3795.
[https://doi.org/10.20546/ijcmas.2018.707.439]

-
Brito LF, Bedere N, Douhard F, Oliveira HR, Arnal M, Peñagaricano F, Schinckel AP, Baes CF and Miglior F. (2021). Review: Genetic selection of high-yielding dairy cattle toward sustainable farming systems in a rapidly changing world. Animal. 15:100292.
[https://doi.org/10.1016/j.animal.2021.100292]

-
Bruce BB and Boateng ID. (2025). Pineapple by-products: A critical review of their bioactive compounds as eco-friendly pesticides in pest management. Food Chemistry: X. 28:102567.
[https://doi.org/10.1016/j.fochx.2025.102567]

-
Chen Y, Chen X, Baserdem B, Zhan H, Li Y, Davis MB, Kebschull JM, Zador AM, Koulakov AA and Albeanu DF. (2022). High-throughput sequencing of single neuron projections reveals spatial organization in the olfactory cortex. Cell. 185:4117-4134.e28.
[https://doi.org/10.1016/j.cell.2022.09.038]

-
Dhiman S, Kaur S, Thakur B, Singh P and Tripathi M. (2025). Nutritional enhancement of plant-based fermented foods: Microbial innovations for a sustainable future. Fermentation. 11:346. https://www.mdpi.com/2311-5637/11/6/346, (cited by 2025 Dec 17).
[https://doi.org/10.3390/fermentation11060346]

-
Ghaffari MH, Rezaei-Ahvanooei MR, Piray AH, Bahrampour J, Ma T and Bradford BJ. (2025). Effects of rumen-protected choline supplementation on lactation performance of dairy cows: A systematic review and dose-response meta-analysis. Journal of Dairy Science, 108:9516-9534.
[https://doi.org/10.3168/jds.2024-25844]

-
Gowda NK, Vallesha NC, Awachat VB, Anandan S, Pal DT and Prasad CS. (2015). Study on evaluation of silage from pineapple(Ananas comosus) fruit residue as livestock feed. Tropical Animal Health and Production. 47:557-561.
[https://doi.org/10.1007/s11250-015-0762-2]

-
Habotta OA, Dawood MAO, Kari ZA, Tapingkae W and Van Doan H. (2022). Antioxidative and immunostimulant potential of fruit derived biomolecules in aquaculture. Fish & Shellfish Immunology. 30:317-322.
[https://doi.org/10.1016/j.fsi.2022.09.029]

-
Hawanis HSN, Ilyas RA, Ahmad MH, Xin CH, Mahadi NKI, Atiqah A, Kaur K, Sapuan SM, Muhammad D, Saharudin MS and Petrů M. (2025). Recent developments in pineapple (Ananas comosus L.) based biocomposites and their potential industrial applications: A review. International Journal of Biological Macromolecules. 328:147531.
[https://doi.org/10.1016/j.ijbiomac.2025.147531]

-
Jang KB, Purvis JM and Kim SW. (2020). Supplemental effects of dietary lysophospholipids in lactation diets on sow performance, milk composition, gut health, and gut-associated microbiome of offspring. Journal of Animal Science. 98.
[https://doi.org/10.1093/jas/skaa227]

-
Kalogerakou T and Antoniadou M. (2024). The role of dietary antioxidants, food supplements and functional foods for energy enhancement in healthcare professionals. Antioxidants. 13:1508. https://www.mdpi.com/2076-3921/13/12/1508, (cited by 2025 Dec 17).
[https://doi.org/10.3390/antiox13121508]

-
Ketnawa S, Chaiwut P and Rawdkuen S. (2011). Extraction of bromelain from pineapple peels. Food Science and Technology International. 17:395-402.
[https://doi.org/10.1177/1082013210387817]

-
Ketnawa S, Chaiwut P and Rawdkuen S. (2012). Pineapple wastes: A potential source for bromelain extraction. Food and Bioproducts Processing. 90:385-391.
[https://doi.org/10.1016/j.fbp.2011.12.006]

-
Khan MZ, Huang B, Kou X, Chen Y, Liang H, Ullah Q, Khan IM, Khan A, Chai W and Wang C. (2023). Enhancing bovine immune, antioxidant and anti-inflammatory responses with vitamins, rumen-protected amino acids, and trace minerals to prevent periparturient mastitis. Frontiers in Immunology. 14:1290044.
[https://doi.org/10.3389/fimmu.2023.1290044]

-
Kholif AE, Hassan AA, El Ashry GM, Bakr MH, El-Zaiat HM, Olafadehan OA, Matloup OH and Sallam SMA. (2021). Phytogenic feed additives mixture enhances the lactational performance, feed utilization and ruminal fermentation of Friesian cows. Animal Biotechnology. 32:708-718.
[https://doi.org/10.1080/10495398.2020.1746322]

-
Kyawt YY, Win KS, Mu KS, Aung A and Aung M. (2020). Feeding pineapple waste silage as roughage source improved the nutrient intakes, energy status and growth performances of growing Myanmar local cattle. Journal of Advanced Veterinary and Animal Research. 7:436-441.
[https://doi.org/10.5455/javar.2020.g439]

-
Mehraj M, Das S, Feroz F, Waheed Wani A, Dar SQ, Kumar S, Wani AK and Farid A. (2024). Nutritional composition and therapeutic potential of pineapple peel - A comprehensive review. Chemistry & Biodiversity. 21:e202400315.
[https://doi.org/10.1002/cbdv.202400315]

- National Research Council. (2001). National Research Council (NRC). Nutrient Requirements of Dairy Cattle, 7th, Washington DC.
-
Polanía AM, Londoño L, Ramírez C, Bolivar G and Aguilar CN. (2023). Valorization of pineapple waste as novel source of nutraceuticals and biofunctional compounds. Biomass Conversion and Biorefinery. 13:3593-3618.
[https://doi.org/10.1007/s13399-022-02811-8]

-
Sahakyan N. (2026). Plant-derived phenolics as regulators of nitric oxide production in microglia: Mechanisms and therapeutic potential. Medical Gas Research. 16:169-175.
[https://doi.org/10.4103/mgr.MEDGASRES-D-25-00058]

-
Santos NW, Santos GTD, Silva-Kazama DC, Grande PA, Pintro PM, de Marchi FE, Jobim CC and Petit HV. (2014). Production, composition and antioxidants in milk of dairy cows fed diets containing soybean oil and grape residue silage. Livestock Science. 159:37-45.
[https://doi.org/10.1016/j.livsci.2013.11.015]

-
Seo YS, Ryu SM, Lee J, Jeong H, Choi G, Moon BC, Lim JO, Nam HH, Kim JS and Lee S. (2025). Protective effect of cast-off skin of cicadidae periostracum water extract in a radiation-induced testicular injury mice model. Food Science & Nutrition. 13:e70198.
[https://doi.org/10.1002/fsn3.70198]

-
Somagond YM, Das P, Dang AK, Yadav DK, Kittur PM, Panda BS K, Devi P, Kamboj A and Alhussien MN. (2025). Parenteral micronutrient supplementation enhances mammary immune function and colostrum-milk quality by modulating cytokine profiles and oxidative stress in transition crossbred cows. Frontiers in Immunology. 16:1669246.
[https://doi.org/10.3389/fimmu.2025.1669246]

-
Sukri SAM, Andu Y, Sarijan S, Khalid HNM, Kari ZA, Harun HC, Rusli ND, Mat K, Khalif RIAR, Wei LS, Rahman MM, Hakim AH, Norazmi Lokman NH, Hamid NKA, Khoo MI and Doan HV. (2023). Pineapple waste in animal feed: A review of nutritional potential, impact and prospects. Annals of Animal Science. 23:339-352.
[https://doi.org/10.2478/aoas-2022-0080]

-
Van Soest PJ, Robertson JB and Lewis BA. (1991). Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. Journal of Dairy Science. 74:3583-3597.
[https://doi.org/10.3168/jds.S0022-0302(91)78551-2]

-
Zhu T, Sun L, Li Q, Jiang J, Zhang H, Ye Z, Chen J and Guo J. (2025). Effects of exogenous protease supplementation on growth performance, liver health, immunity and intestinal microbiota of grass carp(Ctenopharyngodon idella). Aquaculture Reports. 43:102878.
[https://doi.org/10.1016/j.aqrep.2025.102878]