Morphophysiological Responses of Two Mustard (Brassica juncea L.) Cultivars to Foliar Application of Gibberellic Acid (GA3) and Qualitative Analysis of Major Flavonol Glycosides
This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Mustard (Brassica juncea L.) is a medicinal oilseed crop of the Brassicaceae family, globally used as a food spice and folk medicine. Although extensive research has already been conducted on gibberellin (GA) application to mustard, studies focusing on specific cultivars such as ‘Asia Jeok’, used as a leafy vegetable and kimchi ingredient, and ‘Red Frill’, used as a spice, remain limited. Therefore, in this study, we investigated the morphophysiological responses of these two cultivars to GA treatment and performed a qualitative analysis of flavonol glycosides among their bioactive compounds.
We performed a single foliar application of GA3 at 0 (control), 50, 100, 150, or 200 mg·ℓ-1 to four-week-old seedlings of two cultivars, ‘Asia Jeok’ (AJ) and ‘Red Frill’ (RF), and evaluated their morphological traits, biomass accumulation and allocation, plant quality indices, remote sensing-based vegetation indices, and OJIP chlorophyll fluorescence parameters four weeks later. GA3 promoted shoot width and leaf expansion in both cultivars, with the strongest responses generally observed at 50 mg·ℓ-1. Thereafter, cultivar responses diverged. In AJ plants, GA3 at 200 mg·ℓ-1 enhanced shoot biomass and yielded higher values for compactness and Dickson quality index, indicating coordinated growth and structural quality improvements. In RF plants, morphological performance was most favorable at 50 mg·ℓ-1, whereas treatment with 150 mg·ℓ-1 GA3 coincided with higher energy dissipation and lower performance index on an absorption basis than those in the control, suggesting a shift toward less favorable photochemical adjustment at intermediate-to-high concentrations. In addition, chemical profiling through UHPLC-PDA-ESI-MS/MS identified 11 major flavonol glycosides with distinct dominant cultivar-specific peaks.
Our results support cultivar-specific GA3 application to mustard crops and indicate that intermediate-to-high concentrations might unfavorably alter photochemistry.
Keywords:
Brassica juncea, Biomass Allocation, Chlorophyll Fluorescence, Flavonol Glycosides, OJIP Transient, Photosystem II, Plant Growth Regulator, Remote Sensing Vegetation IndicesINTRODUCTION
Recent advances in crop production have increasingly underscored the importance of growth management strategies that enable precise, stage-specific regulation of plant traits and reliable attainment of desired production targets (Gebbers and Adamchuk, 2010; Rezaei et al., 2017). Within this framework, plant growth regulators (PGRs) have been used as practical tools to directly modulate plant growth and development (Rademacher, 2015; Lee et al., 2025a), and their application has been evaluated across a wide range of crops under specific management objectives (Rajala, 2004; Lee et al., 2020; Lee et al., 2024; Wu et al., 2024).
Among the PGRs, gibberellins (GAs) are representative phytohormones that regulate multiple growth and developmental processes, including shoot elongation, leaf expansion, and the transition to reproductive development, and exogenous application is well documented to elicit pronounced changes in plant morphology (Hauvermale et al., 2012; Davière and Achard, 2013). In addition to morphological responses, gibberellic acid (GA3) treatment can also be accompanied by shifts in photosynthetic performance, plant water status, biomass accumulation, and resource allocation patterns (Iqbal et al., 2011; Fu et al., 2023). Accordingly, a rigorous assessment of the effects of GA3 would be facilitated by an integrated morphophysiological framework that considers physiological indicators along with growth and allocation traits.
In this framework, GA3-induced morphological expansion and biomass allocation are expected to alter canopy structure and light interception (Hedden and Thomas, 2012), thereby potentially influencing PSII energy partitioning, as reflected by chlorophyll fluorescence (Kramer et al., 2004). In addition, cultivar-specific antioxidant/photoprotective metabolite backgrounds may modulate physiological stability under rapid growth stimulation (Agati et al., 2013); thus, qualitative metabolite profiling can provide a supportive baseline context for interpreting cultivar-dependent photochemical responses to GA3.
Mustard (Brassica juncea L.) is an economically important vegetable and oil crop with recognized nutraceutical and medicinal relevance. Mustard plants have been traditionally used for the treatment of various diseases, including cancer, obesity, depression, and diabetes. Furthermore, diverse biological activities, including anticancer, antioxidant, antiviral, and antidiabetic effects, have been reported, attributable to bioactive phytochemicals such as glucosinolates and phenolic compounds (Tian and Deng, 2020; Zhang et al., 2023; Hu and Yan, 2025).
Interestingly, B. juncea cultivars exhibit substantial diversity in morphological traits and growth habits (Vinu et al., 2013). Even under identical PGR regimes, responses can differ as a function of genetic background and cultivar-specific characteristics (McCabe and Burke, 2021; Dick and VanderWeide, 2025). More broadly, such cultivar-level variation can extend to responsiveness to exogenous hormone applications, such that the morphophysiological outcomes elicited by the same GA3 concentration may differ among cultivars (Pavlista et al., 2012; Elahi et al., 2022). Therefore, an experimental comparison of cultivar-specific responses to the foliar application of GA3 is warranted.
Foliar application is operationally straightforward and allows for a relatively flexible selection of application timing and concentration, making it a practical approach for inducing and regulating target traits in a stage-specific manner (Niu et al., 2021). Direct delivery through the leaf surface is highly amenable to field applications, and both the application intensity and frequency can be adjusted in a management-oriented fashion to accommodate the prevailing production conditions, thereby facilitating a stepwise growth management strategy (Noack et al., 2010; Lovatt, 2013).
To evaluate the effects of foliar-applied GA3 from a physiological perspective, employing metrics that are suitable for use in the field is important. Chlorophyll fluorescence provides a non-destructive (or non-invasive) proxy for photosystem II (PSII) photochemical efficiency and stress-related responses (Jang et al., 2023), enabling sensitive detection of treatment-associated shifts in photochemical performance (Maxwell and Johnson, 2000; Kim et al., 2024a). In addition, remote sensing-based vegetation indices are useful for inferring the pigment status and optical properties of leaves and thus can support the rapid assessment of plant conditions in situ (Xue and Su, 2017; Lee et al., 2025b). Collectively, the integration of these approaches is expected to strengthen the interpretation of physiological responses to exogenous GA3 and provide an empirical basis for linking morphological and physiological responses within a unified framework.
The physiological effects of GA foliar application are variable across mustard cultivars. Composition and content of bioactive compounds also show variation between cultivars (Arena et al., 2020; Ibrahim et al., 2023) and those differences may be critical determinants of plant quality and medicinal value of mustard crops. Although B. juncea is primarily known as an oilseed crop, its aerial parts are widely consumed as food and used as medicine, such as in ‘ssam’ (wraps) or ‘kimchi’, particularly in Korea. Therefore, the identification and quantification of bioactive components in the aerial parts of different mustard cultivars are beneficial for evaluating their medicinal value. Despite general acknowledgement of biochemical differences among mustard cultivars, studies in which the composition and content of these compounds have been compared across cultivars are limited.
Accordingly, we not only examined the effects of gibberellin on the morphophysiological characteristics of different cultivars but also performed a qualitative analysis of bioactive compounds in each cultivar. The results of this study serve as a basic reference to support future quantitative evaluations of bioactive compounds following GA treatment. Given that functional secondary metabolites, including glucosinolates and flavonoids, have been reported in B. juncea, our qualitative analysis specifically focused on flavonol glycosides as a major compound group closely associated with antioxidant activity (Heim et al., 2002; Ahn et al., 2007; Huang et al., 2022).
Thus, the tandem objectives of this study were to a) quantify the key morphological traits and physiological responses (specifically, chlorophyll fluorescence and vegetation indices) of two mustard cultivars in response to foliar-applied GA3 and b) provide complementary chemical baseline information by conducting UHPLC-PDA-ESI-MS/MS-based qualitative profiling to identify the major flavonol glycoside peaks in the two cultivars.
MATERIALS AND METHODS
1. Preparation of plant materials
Seeds of two mustard (B. juncea) cultivars, ‘Asia Jeok’ (AJ) and ‘Red Frill’ (RF), were obtained from a seed company (Asia Seed, Seoul, Korea). A propagation medium was prepared by mixing a non-fertilized horticultural substrate (Hanareumsangto, Shinsung Mineral, Goesan-gun, Korea), perlite (Ecolite Perlite, Homansaneob, Jeongeup, Korea), and vermiculite (Ecolite Jilseog, Homansaneob, Jeongeup, Korea) at a 1:1:1 (v/v/v) ratio, and the mixture was filled into 105-cell plug trays. Seeds were sown at three seeds per cell.
After sowing, the trays were transferred to a closed nursery system in an experimental greenhouse at the Department of Environmental Horticulture, Sahmyook University (Seoul, Korea). The seedlings were grown on nursery benches (1.2 × 0.7 × 0.6 m; width × length × height). White light-emitting diodes at color temperature 4100 K (T5 LED, Zhong Shan Jinsung Electronic, Zhōngshān, China) were used as the light source, and photosynthetic photon flux density was maintained at 100 μmol·m-2·s-1. During the nursery period, air temperature and relative humidity were maintained at 20 ± 1℃ and 62.3 ± 16.9%, respectively. Following the method described by Lee et al. (2025b), a nutrient solution was supplied twice weekly via sub-irrigation. The nutrient solution was prepared by dissolving 15 g of a 4-18-38 premix (Masterblend International-Tyler Enterprises, Morris, IL, USA), 7.5 g of MgSO4 (Smartro MgS, Busan, Korea), and 15 g of Ca(NO3)2 (Smartro CAL, Gijang-gun, Busan, Korea) in 20 ℓ of purified water. Approximately four weeks after sowing, the seedlings were thinned to one uniformly developed plant per cell. The selected seedlings were then hardened for 72 h prior to use in the experiment.
2. Experimental design and environmental location
The field experiment was conducted at the Sahmyook Green Education Practice Center, Sahmyook University, Seoul, Korea (37°38’16”N, 127°06’30”E) for four weeks, from September 16 to October 15, 2024. Typically, leaf mustard is harvested 35 to 40 days after sowing in the summer and 60 to 70 days in the autumn and winter seasons (Assefa et al., 2023). Several studies on leaf mustard have evaluated morphophysiological traits by harvesting plants 28 to 35 days after transplanting seedlings. Accordingly, we selected four weeks after transplantation as the time point for evaluating the responses to GA3 treatment (Maršić et al., 2021; Janah et al., 2023).
Exogenous GA3 (CAS No. 77-06-5; Sigma-Aldrich, St. Louis, MO, USA) was applied as a foliar spray at five concentrations: 0 (control), 50, 100, 150, or 200 ㎎·ℓ-1. GA3 was first dissolved in 10 ㎖ of 95% EtOH and then diluted with purified water to the final concentrations. Foliar applications were performed 72 h after transplantation. In each cultivar × GA3 treatment, a total of 1 ℓ of the prepared solution was applied using a hand sprayer, equally distributed across the five replicates (200 ㎖ per replicate), to ensure full coverage of the plant canopies.
The GA3 application rates were based on those used in previous studies of GA3 application to Brassica crops (Elahi et al., 2022; Prodhan et al., 2022; Shahi et al., 2022). The selected range of GA3 concentrations allows for a practical foliar spray concentration window, enabling the assessment of concentration-dependent responses.
During the experimental period, ambient air temperature, relative humidity, and mean cloudiness averaged 27.8 ± 3.8℃, 70.4 ± 13.7%, and 5.4 ± 1.3 okta, respectively. The physicochemical properties of the experimental field soils are listed in Table 1.
3. Parameters and plant quality indices
The following growth- and yield-related parameters were measured: shoot height, shoot width, leaf length, leaf width, number of leaves, leaf area, stem diameter, root length, main root thickness, and the fresh and dry weights of shoots and roots. The chlorophyll content (SPAD units) was determined using a portable chlorophyll meter (SPAD-502Plus, Konica Minolta, Tokyo, Japan). Compactness (Eq. 1), the Dickson quality index (DQI; Eq. 2) and relative moisture content (RMC; Eq. 3) were calculated following Hong et al. (2025), Dickson et al. (1960), and Lee and Nam (2024).
| (Eq. 1) |
| (Eq. 2) |
| (Eq. 3) |
(abbreviation; SDW: shoot dry weight; SH: shoot height; TDW: total dry weight; SD: stem diameter; RDW: root dry weight; FW: fresh weight; and DW: dry weight)
Leaf color characteristics were evaluated using the Commission Internationale de l’Eclairage Lab (CIELAB) color space. A spectrophotometer (CM-2600d, Konica Minolta, Tokyo, Japan) was set to D65/10° and operated in the specular component included (SCI) mode to record L*, a*, and b* values. The measurement conditions and procedures were the same as those described by Lee (2023).
The mean CIELAB coordinates (L*, a*, and b*) for each treatment were converted into their corresponding digital color representations using Converting Colors (Zettl, 2026). The resulting color chips were used to support a qualitative visual assessment of leaf color differences among the treatments.
4. Remote sensing vegetation indices and chlorophyll fluorescence
Remote sensing-based vegetation indices were measured using a portable spectroradiometer (PolyPen RP410; Photon Systems Instruments, Drásov, Czech Republic). The anthocyanin reflectance index 2 (ARI2; Eq. 4), and carotenoid reflectance index 2 (CRI2; Eq. 5), normalized difference vegetation index (NDVI; Eq. 6), photochemical reflectance index (PRI; Eq. 7), and modified chlorophyll absorption ratio index (MCARI; Eq. 8) were calculated using the equations given by Lee et al. (2025b).
| (Eq. 4) |
| (Eq. 5) |
| (Eq. 6) |
| (Eq. 7) |
| (Eq. 8) |
Physiological responses were evaluated by measuring chlorophyll fluorescence using a portable fluorometer (FluorPen FP 110/D; Photon Systems Instruments, Drásov, Czech Republic). Measurements were conducted at night (22:00-04:00) to ensure full dark adaptation in accordance with the manufacturer’s guidelines (PSI, 2026). The measurement protocol was based on that described by Shin et al. (2024). Briefly, the excitation wavelength was set to 455 ㎚ and Fm for the JIP-test was induced by applying saturating pulse at 1,500 μmol·m-2·s-1, corresponding to 50% of the super pulse. The selected fluorescence parameters were Vj (Eq. 9), Vi (Eq. 10), Fv/Fm (Eq. 11), Mo (Eq. 12), PIABS (Eq. 13), ΦPo (Eq. 14), Ψo (Eq. 15), ΦEo (Eq. 16), ΦDo (Eq. 17), ABS/RC (Eq. 18), TRo/RC (Eq. 19), ETo/RC (Eq. 20), and DIo/RC (Eq. 21). The equations, as given by Kim et al. (2024a) and Lee et al. (2025b) are as follows:
| (Eq. 9) |
| (Eq. 10) |
| (Eq. 11) |
| (Eq. 12) |
| (Eq. 13) |
| (Eq. 14) |
| (Eq. 15) |
| (Eq. 16) |
| (Eq. 17) |
| (Eq. 18) |
| (Eq. 19) |
| (Eq. 20) |
| (Eq. 21) |
5. Preparation of samples for UHPLC-PDA-ESI-MS/MS analysis
The leaves of the mustard plants were hot-air dried at 120℃ and then pulverized. Powdered samples (0.5 g) were extracted with 50% MeOH (3 × 5 ㎖ for 1 h each) at 45℃. After the extract was concentrated using a nitrogen evaporator, an appropriate amount of MeOH was added, and the resultant extract solution was filtered to make an HPLC sample with a concentration of 1 ㎎·㎖-1.
6. UHPLC-PDA-ESI-MS/MS analysis
The total extracts from the mustard plants were analyzed using a UHPLC system (Vanquish Flex; Thermo Fisher Scientific, Waltham, MA, USA) coupled with a quadrupole time-of-flight (qTOF) mass spectrometer (Q-TOF 5600; AB SCIEX, Framingham, MA, USA) and a photodiode array detector (Ultimate 3000 PDA detector; Thermo Fisher Scientific, Waltham, MA, USA). Liquid chromatographic analyses were performed on a C18 reverse-phase LC column (Waters Cortecs T3, 1.6 ㎛, 2.1 × 150 ㎜). The mobile phase conditions are listed in Table 2. The temperatures for the column oven and sample controller were kept at 45℃ and 10℃, respectively. The volume of each sample injected was 1 ㎕. To ensure efficient and accurate identification of major flavonoids, the ultraviolet (UV) wavelength was set to 330 ㎚.
7. Data analysis
Statistical analyses were performed using SAS 9.4 (SAS Institute, Cary, NC, USA). The experiment was arranged in a completely randomized design (CRD) with five replicates per cultivar × treatment (GA3 concentration). Within each replicate, six plants were measured and averaged to obtain a replicate mean, which was used for statistical analysis (n = 5). Thirty plants were assessed for each treatment combination. The main factors were cultivar and treatment, and a two-way analysis of variance (ANOVA) was conducted to evaluate the effects of these factors and their interaction. Post-hoc comparisons were performed using Duncan’s Multiple Range Test (DMRT) at p < 0.05. Although Tukey’s Honestly Significant Difference (HSD) is effective in controlling Type I errors, it is often considered too conservative for the analysis of data in agricultural studies, potentially increasing the risk of Type II errors (missing true differences). Therefore, DMRT was selected to maximize the statistical power for detecting significant differences among treatments (Carmer and Swanson, 1971).
RESULTS
1. Analysis of morphological traits, plant quality indices, and chlorophyll content
The two mustard cultivars examined in this study responded differently to foliar application of GA3 at different concentrations (Fig. 1 and Fig. 2). At four weeks after foliar GA3 application, shoot height and width varied significantly among treatments (p < 0.001), whereas leaf length and width were affected at p < 0.01 and p < 0.05, respectively. Similarly, leaf number differed significantly among treatments (p < 0.01). In contrast, chlorophyll content (SPAD units) did not differ significantly among the treatments.

Representative photographs of mustard plants (B. juncea) at four weeks after foliar application of gibberellic acid (GA3) at different concentrations.(A) B. juncea cv. Asia Jeok. (B) B. juncea cv. Red Frill.
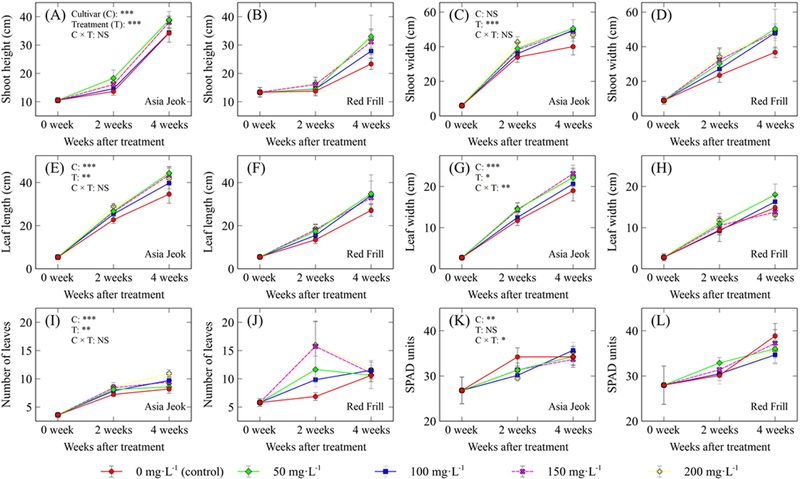
Plant morphological traits and chlorophyll content (SPAD units) of two mustard (B. juncea) cultivars (‘Asia Jeok’ and ‘Red Frill’) measured at four weeks after foliar application of gibberellic acid (GA3) at different concentrations.Panels show (A, B) shoot height, (C, D) shoot width, (E, F) leaf length, (G, H) leaf width, (I, J) number of leaves, and (K, L) chlorophyll content (SPAD units), with the left panel of each pair representing ‘Asia Jeok’ and the right panel representing ‘Red Frill’. Values are means ± standard deviation (SD), (n = 5). NS, non-significant; *, **, and *** indicate significance at p < 0.05, 0.01, and 0.001, respectively (at four weeks after treatment).
In a cultivar-specific comparison, the AJ cultivar exhibited the greatest shoot height at 200 ㎎·ℓ-1 (38.84 ㎝), whereas the RF cultivar reached its maximum shoot height at 50 ㎎·ℓ-1 (33.03 ㎝). Shoot width was maximized at 50 ㎎·ℓ-1 in both cultivars, reaching 50.54 ㎝ in AJ and 50.33 ㎝ in RF. Leaf length and leaf width followed a similar pattern, with the highest mean values observed at 50 ㎎·ℓ-1. Leaf number peaked at 200 ㎎·ℓ-1, with mean values of 10.9 and 11.6 leaves in AJ and RF, respectively.
As shown in Table 3, stem diameter in AJ increased under GA3 treatments of 50-200 ㎎·ℓ-1, ranging from 1.36 to 1.41 ㎝, whereas in RF the thickest stems were obtained at 150 ㎎·ℓ-1 (1.14 ㎝), which was greater than the corresponding value in the control (0.79 ㎝). Ground cover expanded markedly in both cultivars at GA3 concentrations ranging from 50 to 200 ㎎·ℓ-1 and was significantly greater than that of the control.
Plant morphological traits, compactness, and Dickson quality index (DQI) of two mustard (B. juncea) cultivars (‘Asia Jeok’ and ‘Red Frill’) at four weeks after foliar application of gibberellic acid (GA3) at different concentrations.
Leaf thickness did not differ significantly among the treatments in AJ; however, RF exhibited the greatest leaf thickness in the control (0.80 ㎜). Leaf area responses to GA3 differed by cultivar and concentration: in AJ, all GA3 treatments from 50 to 200 ㎎·ℓ-1 resulted in larger leaf areas (824.1-1011.2 ㎠) than was found in the control (665.2 ㎠), whereas in RF, the largest leaf area occurred at 50 ㎎·ℓ-1 (643.1 ㎠), compared with 408.2 ㎠ in the control.
Root length did not differ significantly among treatments in AJ, whereas in RF the longest roots were obtained at 50 ㎎·ℓ-1 GA3 (14.90 ㎝), exceeding those at 100 ㎎·ℓ-1 (9.69 ㎝). Main root thickness in AJ was the greatest at 150-200 ㎎·ℓ-1 (1.48-1.53 ㎝), whereas in RF, GA3 treatments of 50-200 ㎎·ℓ-1 increased main root thickness to 0.81-0.90 ㎝ relative to the control (0.45 ㎝).
With respect to plant quality indices, compactness in AJ was the highest at 200 ㎎·ℓ-1 GA3 (0.645), exceeding the control (0.380), whereas compactness in RF did not differ significantly among treatments. As for DQI, the AJ cultivar showed higher values than did the control (0.271) at 50, 150, and 200 ㎎·ℓ-1 GA3 (0.417, 0.457, and 0.521, respectively). In the RF cultivar, DQI was relatively higher at 150 ㎎·ℓ-1 (0.328) than in the control (0.128).
2. Analysis of biomass and relative moisture content
Regarding biomass, shoot fresh weight increased in both cultivars at 200 ㎎·ℓ-1 GA3, reaching 379.7 g in AJ and 157.0 g in RF, compared with the respective values in the controls (202.5 and 73.7 g) (Table 4). Shoot dry weight in AJ was also highest at 200 ㎎·ℓ-1 (25.09 g), exceeding the control (13.19 g). However, shoot dry weight in RF did not differ significantly among treatments.

Biomass components (fresh and dry weights) and relative moisture content of two mustard (B. juncea) cultivars (‘Asia Jeok’ and ‘Red Frill’) at four weeks after foliar application of gibberellic acid (GA3) at different concentrations.
Root fresh weight in AJ was greater under 150-200 ㎎·ℓ-1 GA3 (10.94-13.43 g) than under lower concentrations. In RF, root fresh weight increased across 50-200 ㎎·ℓ-1 (5.14-6.38 g) relative to the control (1.24 g). Root dry weight in AJ ranged from 0.97 to 1.11 g at 150-200 ㎎·ℓ-1, which was higher than the corresponding value in the control (0.65 g), but was not significantly different from the values observed at 50 and 100 ㎎·ℓ-1 (0.96 and 0.88 g, respectively). In RF, root dry weight under 50-200 ㎎·ℓ-1 ranged from 0.88 to 0.93 g, which was greater than the control (0.27 g).
The relative moisture content of the shoots did not differ significantly among treatments in either cultivar. In contrast, in AJ, the relative moisture content of the root decreased at 50-100 ㎎·ℓ-1 compared with the control, whereas the values at 150-200 ㎎·ℓ-1 were statistically comparable to the control. In RF, the relative moisture content of the root was significantly higher under 50-200 ㎎·ℓ-1 (83.3-84.9%) than in the control group (77.3%).
3. Analysis of shoot external quality and leaf pigments
Among external quality attributes, the CIELAB lightness coordinate (L*) did not differ significantly among treatments in AJ; however, in RF, L* was highest at 200 ㎎·ℓ-1 GA3 (28.03) (Table 5). In AJ, the a* coordinate (green–red opponent axis) showed a pattern similar to L*, with no significant differences among treatments. In RF, however, this pattern was not observed; a* was greatest in the control (5.72) and lowest at 200 ㎎·ℓ-1 (3.99). As for the b* coordinate (blue-yellow opponent axis), AJ exhibited a higher value at 150 ㎎·ℓ-1 (5.70) than at 200 ㎎·ℓ-1 (1.15), whereas RF showed the highest b* at 200 ㎎·ℓ-1 (3.52) relative to the other treatments.

CIELAB color coordinates and pigment-related vegetation indices of two mustard (B. juncea) cultivars (‘Asia Jeok’ and ‘Red Frill’) at four weeks after foliar GA3 application at different concentrations, including anthocyanin reflectance index 2 (ARI2) and carotenoid reflectance index 2 (CRI2).
Among the remote sensing-based vegetation indices, anthocyanin reflectance index 2 (ARI2), used as a proxy for leaf anthocyanin status, did not differ significantly among the treatments in either cultivar. Similarly, the carotenoid reflectance index 2 (CRI2), an index associated with carotenoid-related reflectance characteristics, showed no significant treatment effects in either cultivar.
4. Analysis of remote sensing vegetation indices and chlorophyll fluorescence responses
With respect to the remote sensing-based vegetation indices, the NDVI, an indicator of overall plant vigor, tended to be higher in the 10 0 and 20 0 ㎎·ℓ-1 GA3 treatments than in the control in AJ and RF, respectively (Table 6). Specifically, NDVI increased from 0.694 (control) to 0.716 at 100 ㎎·ℓ-1 in AJ, and from 0.703 (control) to 0.726 at 200 ㎎·ℓ-1 in RF. The PRI, a parameter associated with photochemical efficiency, did not differ significantly among treatments in AJ; however, in RF, PRI increased to 0.008 at both 100 and 200 ㎎·ℓ-1 compared with the result in the control group (0.003). MCARI, which is closely related to the leaf chlorophyll status, did not show significant treatment effects in either cultivar.

Remote sensing vegetation indices of two mustard (B. juncea) cultivars (‘Asia Jeok’ and ‘Red Frill’) at four weeks after foliar application of GA3 at different concentrations, including the normalized difference vegetation index (NDVI), photochemical reflectance index (PRI), and modified chlorophyll absorption ratio index (MCARI), together with technical chlorophyll fluorescence parameters and the performance index on an absorption basis (PIABS).
In terms of chlorophyll fluorescence parameters, relative variable fluorescence at the J-step (Vj) was higher in AJ under 150-200 ㎎·ℓ-1 GA3 (0.292-0.297), and in RF it was the highest at 150 ㎎·ℓ-1 (0.307) relative to the control (0.266). Relative variable fluorescence at the I-step (Vi) did not differ significantly among treatments in AJ, whereas in RF, Vi was higher across 50-200 ㎎·ℓ-1 (0.587-0.601). The maximum quantum yield of PSII (Fv/Fm) in AJ reached 0.854 at 100 ㎎·ℓ-1, which was significantly higher than the value at 200 ㎎·ℓ-1 (0.849). In RF, Fv/Fm remained relatively high in the control and under 50-100 ㎎·ℓ-1 (0.848-0.849), but decreased to 0.844 at 150 ㎎·ℓ-1, indicating a statistically lower mean. The initial slope of the fluorescence induction curve (Mo) was relatively elevated in AJ under 150-200 ㎎·ℓ-1 (0.336-0.340), whereas in RF, Mo increased to 0.356 at 150 ㎎·ℓ-1 compared with the control (0.265).
The performance index on an absorption basis (PIABS) in AJ was higher under 50-100 ㎎·ℓ-1 (12.86-13.13) than under 200 ㎎·ℓ-1 (10.51). In RF, PIABS was the highest in the control, with a value of 13.56.
According to the chlorophyll fluorescence parameters associated with quantum yields, foliar application of GA3 elicited a broad spectrum of physiological responses, and all analyzed variables showed significant treatment effects, with p-values ranging from p < 0.01-0.001 (Fig. 3 and Table 7). The maximum quantum yield parameter, ΦPo, was identical to the pattern observed for Fv/Fm. The probability that a trapped exciton drives electron transfer beyond QA into the electron transport chain (Ψo) was the highest in the AJ cultivar under 50 ㎎·ℓ-1 (0.733) and in the RF cultivar in the control (0.733). Similarly, the quantum yield of electron transport (ΦEo) reached its maximum in AJ at 50 ㎎·ℓ-1 (0.619), whereas RF exhibited the highest ΦEo in the control (0.623). By contrast, the quantum yield of energy dissipation (ΦDo), representing the probability that absorbed excitation energy is dissipated, was the highest in AJ at 200 ㎎·ℓ-1 (0.150) and in RF at 150 ㎎·ℓ-1 (0.155).
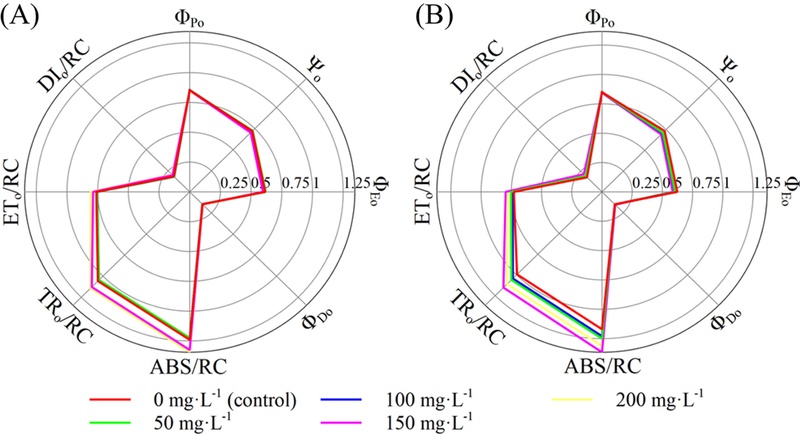
Chlorophyll fluorescence-derived quantum efficiencies and specific energy flux indices of two mustard (B. juncea) cultivars (‘Asia Jeok’ and ‘Red Frill’) at four weeks after foliar application of GA3 at different concentrations.(A, B) quantum efficiency parameters and specific energy flux indices of mustard cultivar ‘Asia Jeok’ and ‘Red Frill’, respectively.

Significance levels for OJIP fluorescence-derived parameters in two mustard (B. juncea) cultivars (‘Asia Jeok’ and ‘Red Frill’) measured four weeks after foliar application of GA3 at different concentrations, including quantum efficiencies (ΦPo, Ψo, ΦEo, and ΦDo) and specific energy fluxes per reaction center (ABS/RC, TRo/RC, ETo/RC, and DIo/RC).
With respect to specific energy fluxes per reaction center (RC), ABS/RC, which reflects the absorption flux per RC, was the greatest in AJ at 200 ㎎·ℓ-1 and in RF at 150 ㎎·ℓ-1 (1.34 and 1.36, respectively). The trapped energy flux per RC (TRo/RC), electron transport flux from QA to QB per RC (ETo/RC), and dissipated energy flux per RC (DIo/RC) exhibited the same overall patterns as those of ABS/RC.
5. Qualitative analysis of total extracts of B. juncea using UHPLC-PDA-ESI-MS/MS
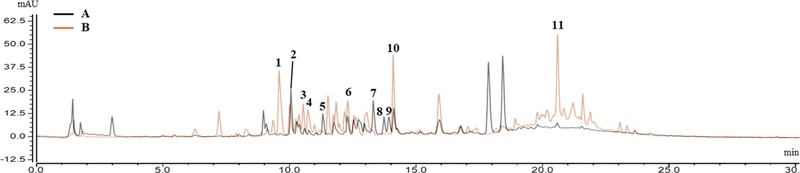
Based on the UV chromatogram at 330 ㎚, a representative wavelength that flavonoids absorb, major peaks were surveyed for their mass spectra with the key product MS2 ions in positive mode (Table 8). The mass spectrum of compound 2 exhibited a protonated ion [M + H]+ at m/z 773.2164 and key diagnostic product MS2 ions at m/z 611.1647, 449.1087, and 287.0564. The MS2 ion at m/z 287.0564 indicated the presence of kaempferol as an aglycone. Starting with the outermost substituents of the molecule, the MS2 ions at m/z 611.1647 and 449.1087 were likely formed by detachment of a glucosyl group at C-7 (m/z difference: 162.0517) and an additional glucosyl group at C-3 (m/z difference: 162.0560).

UHPLC-PDA-ESI MS/MS data and putative identification of major flavonol glycosides in two cultivars (‘Asia Jeok’ and ‘Red Frill’) of mustard (B. juncea).
Compound 5 was determined to be a flavonol glycoside with hydroxycinnamic acid (HCA) derivatives; the mass spectrum showed MS2 ions at m/z 611.1625, indicating the detachment of one glucose unit at C-7 (m/z difference: 162.0552) and subsequent hydroxyferulic acid (m/z difference: 192.0410). The presence of hydroxyferulic acid was supported by a UV absorption maximum at 330.5 ㎚ in additional UV analysis. Similarly, the remaining compounds were tentatively identified, as shown in Table 8 and Figure 4. Interestingly, compounds 2, 5, 7, 8, and 9 were found to be the major flavonol glycosides in AJ, whereas compounds 1, 3, 4, 6, 10, and 11 were the major flavonol glycosides in RF (Fig. 5).
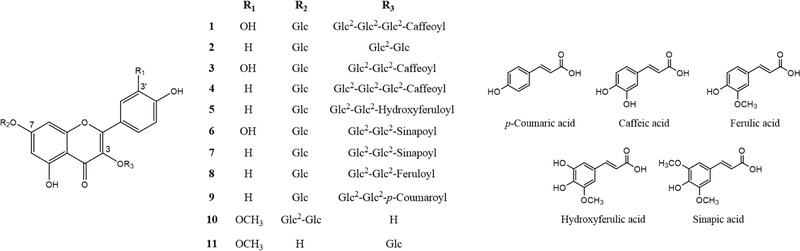
Structures of major flavonoid glycosides from two cultivars (‘Asia Jeok’ and ‘Red Frill’) of mustard (B. juncea).
DISCUSSION
GAs are among the principal phytohormones governing morphogenetic processes during the vegetative growth stage (Yamaguchi, 2008). Responses to exogenous GA3 application can vary depending on species- and cultivar-specific sensitivity, as well as the applied concentration, and GA3-induced morphological changes may, in turn, trigger cascading effects on biomass accumulation, resource allocation patterns, and broader physiological traits (Pavlista et al., 2012; Shaddad et al., 2013; Rademacher, 2015).
In the present study, we conducted an integrated assessment of the effects of foliar-applied GA3 on two mustard cultivars, including morphological responses, biomass accumulation and partitioning, external quality attributes, and physiological responses, which were quantified using chlorophyll fluorescence and remote sensing-based vegetation indices. Overall, our results indicate that GA3 treatment elicited significant changes in several morphological traits and selected photochemical parameters, with the magnitude and significance depending on cultivar, GA3 concentration, and the parameter evaluated.
Four weeks after foliar GA3 application, shoot height and width differed significantly among the treatments; leaf length, width, and number also showed significant treatment-dependent variation. These findings are consistent with those reported in previous studies, which demonstrated that foliar GA3 application can promote cell elongation in leaves and stems, thereby inducing visually discernible morphological changes within a relatively short time frame (Wenzel et al., 2000; Robil et al., 2025).
Notably, shoot width, leaf length, and leaf width exhibited relatively high mean values in both cultivars under the 50 ㎎·ℓ-1 treatment. This pattern suggests that, with respect to horizontal expansion, a comparatively low GA3 concentration (50 ㎎·ℓ-1) may be effective for the two cultivars examined here, whereas higher concentrations tended to be associated with a modest reduction in plant size. Collectively, these results imply a nonlinear concentration-response relationship of growth parameters with GA3 concentration. Accordingly, an optimum may occur within a certain GA3 concentration range, beyond which supra-optimal concentrations can lead to unfavorable outcomes for specific traits (Tomer, 1984; Islam et al., 2021), highlighting the importance of careful concentration selection.
With respect to shoot height, the AJ cultivar attained its maximum value at 200 ㎎·ℓ-1, whereas the RF cultivar peaked at 50 ㎎·ℓ-1. This cultivar-specific optimum is consistent with previous reports indicating that sensitivity to exogenous GA3 (i.e., responsiveness that may reflect differences in uptake and/or downstream signaling) can vary among cultivars, even within the same species (Muniandi et al., 2018; Ibrahim et al., 2019; Sari, 2024). Taken together, these findings suggest that cultivar-specific recommended concentration ranges should be considered even when the management goal is to improve morphological performance.
In terms of biomass, the AJ cultivar exhibited pronounced increases in shoot fresh weight and shoot dry weight at 200 ㎎·ℓ-1, indicating that relatively high GA3 concentrations promoted assimilate accumulation and overall biomass production in this cultivar. In contrast, RF did not show clear treatment-dependent differences in shoot dry weight, suggesting that despite an increase in apparent plant size following foliar GA3 application, its conversion efficiency from morphological expansion to dry matter accumulation may be comparatively limited relative to AJ.
Regarding belowground traits, both the fresh and dry weight of the roots tended to be higher in the GA3-treated plants than in the control for the RF cultivar. Previous studies have shown that exogenous GA3 can induce morphological changes in shoots and increase biomass accumulation (Castro-Camba et al., 2022). Although the effects of GA are often described as more pronounced in shoots than in roots (Martins et al., 2019; Alam et al., 2022; Lee et al., 2023; Omena-Garcia et al., 2025), our results suggest that GA3-driven leaf expansion and canopy enlargement may have increased light interception and carbon assimilation, which may have translated into enhanced root growth under the conditions of this study.
From the perspective of plant quality indices, the AJ cultivar showed clear improvements in structural quality following foliar GA3 application, as compactness reached its highest value at 200 ㎎·ℓ-1 and DQI exceeded the control at 50, 150, and 200 ㎎·ℓ-1. This pattern indicates that GA3 treatment did not merely stimulate shoot elongation but was accompanied by coordinated changes in traits linked to stem robustness, shoot assimilate accumulation, and shoot-root biomass partitioning. Because the DQI integrates both structural stability and the balance of shoot-to-root allocation (Dickson et al., 1960), these improvements suggest that GA3 application effectively enhances the overall plant quality status in AJ. By contrast, the RF cultivar showed a more limited quality-index response: compactness did not differ significantly among treatments, and DQI was relatively higher at 150 ㎎·ℓ-1 than in the control. Collectively, these results indicate that the extent to which foliar-applied GA3 translates visible morphological responses into integrated quality gains can differ substantially, depending on cultivar identity.
The SPAD units did not differ significantly among treatments. This result indicates that under the conditions applied in the present study, foliar GA3 treatment exerted relatively limited effects on leaf chlorophyll status four weeks after application. Moreover, the primary effects of GA3 appeared to be expressed more strongly through the regulation of morphological and physiological traits than through pigment accumulation, which was supported by the absence of significant treatment effects, not only for SPAD units, but also for ARI2 and CRI2. In a previous study on faba bean (Vicia faba), exogenous GA3 similarly failed to induce significant changes in chlorophyll and carotenoid content (Alam et al., 2022), which is consistent with our observations.
In contrast, NDVI showed an increasing tendency at selected concentrations depending on cultivar (e.g., 100 and 200 ㎎·ℓ-1). Because NDVI is jointly influenced by leaf area and canopy density (Carlson and Ripley, 1997), the GA3-driven increases in ground cover and leaf expansion observed in this study may have contributed to the higher NDVI values. In comparison, MCARI is an index that indirectly reflects chlorophyll status; thus, when treatment effects on SPAD units are limited, the corresponding changes in MCARI may also be constrained. Overall, our results suggest that foliar GA3 application in these mustard cultivars modulated morphological attributes to a greater degree than leaf pigment status. In this context, the present findings align with previous reports indicating that gibberellin effects are often more evident in quantitative growth-related outcomes (e.g., growth and biomass accumulation) than in qualitative traits, such as pigment regulation (Othman et al., 2021; Zhang et al., 2024).
Chlorophyll fluorescence parameters exhibited significant treatment effects for most variables, indicating that GA3 application influenced not only plant morphology, but also energy fluxes within PSII. In particular, treatment-associated variation in quantum-yield-related parameters—ΦPo (Fv/Fm) as well as Ψo, ΦEo, and ΦDo—suggests that foliar GA3 application partially reconfigured PSII photochemical efficiency, the probability of electron transfer beyond QA, and the balance between photochemical energy utilization and energy dissipation (Stirbet and Govindjee, 2011).
Among the two mustard cultivars, the AJ cultivar exhibited the highest Ψo and ΦEo values at 50 ㎎·ℓ-1, suggesting that electron-transport efficiency was comparatively favored under this relatively low GA3 concentration. This pattern is also consistent with the morphological results, in which 50 ㎎·ℓ-1 was advantageous for leaf expansion and increased shoot width, indicating that, in AJ, this concentration may represent a practical range in which improvements in morphological traits and photochemical efficiency can be achieved concurrently. In contrast, the 200 ㎎·ℓ-1 treatment in AJ was associated with an increase in ΦDo, implying a tendency toward a higher fraction of absorbed energy being dissipated rather than utilized photochemically. Nevertheless, considering the values of Fv/Fm, which reflects the maximum quantum yield of PSII, both cultivars maintained relatively high values (0.844-0.854). Although these values were higher than the commonly cited reference range for many healthy, non-stressed higher plants (0.78-0.84) (Muniz et al., 2014; Kim et al., 2024b; Lee et al., 2025b), reference values can vary depending on species, measurement protocol, and instrument settings. Therefore, under the present experimental conditions, the plants were interpreted as not being under pronounced PSII stress, while still showing treatment-related photochemical adjustments in other OJIP fluoresence parameters.
In the RF cultivar, Ψo and ΦEo were relatively high in the control, and PIABS was also greatest in the control. Notably, the 150 ㎎·ℓ-1 treatment was accompanied by an increase in ΦDo, suggesting that, at this concentration, a greater acceptor-side load and/or functional adjustment of PSII reaction centers may have occurred (Stirbet and Govindjee, 2011). Furthermore, the concurrent increases in ABS/RC, TRo/RC, ETo/RC, and DIo/RC at a specific concentration are consistent with the interpretation that a larger fraction of reaction centers shift toward a functionally inactivated state, thereby increasing the apparent energy fluxes per remaining active reaction center (Kim et al., 2024b; Lee et al., 2025b). Collectively, these results imply that meaningful improvement in morphological traits in the RF cultivar is possible under a relatively low GA3 concentration (50 ㎎·ℓ-1), whereas higher concentrations—particularly 150 ㎎·ℓ-1, and to a lesser extent 200 ㎎·ℓ-1—may shift photochemical performance in an unfavorable direction, warranting caution in concentration selection. From a food-crop management perspective, the present results suggest that foliar GA3 application should be managed with cultivar-specific, conservative concentration selection, considering that supra-optimal concentrations can impose a physiological trade-off even when visually discernible growth promotion is observed. Accordingly, applying the minimum concentration required to achieve the desired morphological outcome may be a practical strategy to avoid unintended declines in photochemical efficiency.
In the present study, vegetative growth and PSII performance were assessed four weeks after a single foliar GA3 application; however, GA signaling can exert systemic effects that extend beyond shoot vegetative growth and may influence reproductive sink development and seed oil accumulation in species of Brassica. Foliar application of GA3 has been used in canola to test concentration-dependent shifts in yield components and seed oil content, especially under drought stress (Elahi et al., 2022). Moreover, a field study combined foliar GA3 with mineral nutrition management to evaluate changes in seed yield and oil percentage (Aslam et al., 2023). At the mechanistic level, GA signaling is linked to the transcriptional control of fatty-acid biosynthesis during early seed development, and GA3 treatment of siliques has been shown to increase seed size and induce fatty-acid biosynthetic gene expression (Yan et al., 2021). Therefore, although seed traits were beyond the scope of the present work, future studies should verify whether the concentration window that improved vegetative traits also translates to reproductive performance and oil productivity in these cultivars.
Although the identification of mustard flavonols in this study was initially hampered by challenges in securing reference standards and performing direct isolation, relatively accurate identification was achieved through precise mass determination via HRMS, MS/MS fragmentation pattern analysis, MS library matching, analysis of well-established flavonoid UV patterns, and comparisons with published spectroscopic data for flavonol glycosides. Regarding the aglycone identification, given that kaempferol, quercetin, and isorhamnetin are reported as the predominant aglycones in species of Brassica (Kim et al., 2002; Lin et al., 2011; Neugart and Bumke-Vogt, 2021), excluding the possibility of rhamnetin is reasonable. As for the positioning of the glycosyl groups, a previously published extensive qualitative analysis of phenols in red mustard greens indicates that flavonol 3,7-diglucoside loses its 7-glycosyl group first to form the major MS2 product ion (Lin et al., 2011). Kaempferol 3-sophorotrioside has never been detected in B. juncea, and only a few cases in which triglycosylation occurs at C-3 without glycosylation at C-7 have been reported. These previous results, combined with our finding that compound 2 is one of the major peaks in the AJ cultivar, allowed us to rule out the possibility that compound 2 is kaempferol 3-sophorotrioside.
In summary, qualitative UHPLC-PDA-ESI-MS/MS profiling resulted in the putative identification of 11 major flavonol glycosides, and clear cultivar-dependent differences in the dominant flavonol glycoside peak patterns were observed between the two cultivars. These findings indicate that the composition relative contents of flavonoid glycosides can vary in a cultivar-dependent manner.
Notwithstanding the results of the qualitative analysis, it is difficult to directly correlate GA3 treatment with cultivar-specific variations in flavonol glycoside content in the same way as the morphophysiological changes observed in this study. Nevertheless, the alterations in secondary metabolite content induced by GA3 treatment vary significantly depending on the crop species and cultivar (Park et al., 2017; Sun et al., 2021; Khalil et al., 2023). In light of these findings, conducting quantitative analysis across various mustard cultivars would be useful to elucidate the cultivar-specific effects of GA3 on secondary metabolite biosynthesis.
To summarize the key findings of our study, foliar application of GA3 effectively improved the morphological traits of both mustard cultivars; however, the concentrations of GA3 that resulted in optimal overall growth, plant quality indices, and photochemical responses differed by cultivar. In AJ, the practical optimum depended on the target trait: a relatively low GA3 concentration (50 ㎎·ℓ-1) was favorable for leaf expansion and photochemical performance, whereas a relatively high concentration (200 ㎎·ℓ-1) improved biomass accumulation and DQI. Thus, cultivar-specific recommendations for AJ should be objective-dependent. In contrast, RF tended to exhibit improved morphological traits at a relatively low concentration (50 ㎎·ℓ-1), whereas higher concentrations, particularly 150 ㎎·ℓ-1, may adversely affect photochemical performance.
To build on these findings, we recommend that future studies be designed with the aim of testing alternative application strategies, including GA delivery via root drenches, different timing and frequency of application, and interactions with other plant growth regulators. Lastly, because flavonol glycoside analysis in the current study was limited to qualitative identification, the accumulation of quantitative datasets is necessary to test the relationships between GA3 treatment effects and functional metabolite profiles.
Acknowledgments
This paper was supported by the Sahmyook University Research Fund in 2024. The authors gratefully acknowledge the assistance of Eun Bin Cha, Seungdon Joo, and Jeong Geun Lee (Department of Environmental Horticulture, Sahmyook University, Seoul, Korea) with morphophysiological data measurements. UV chromatogram and additional MS spectrum data were kindly provided by Dr. Doohee Lee (National Instrumentation Center for Environmental Management, Seoul National University, Seoul, Korea).
References
- Ahn BG, Hur JM and Park JC. (2007). Isolations of flavonoids and a higher alcohol from the aerial parts of Brassica juncea. Korean Journal of Pharmacognosy. 38:254-257.
-
Agati G, Brunetti C, Di Ferdinando M, Ferrini F, Pollastri S and Tattini M. (2013). Functional roles of flavonoids in photoprotection: New evidence, lessons from the past. Plant Physiology and Biochemistry. 72:35-45.
[https://doi.org/10.1016/j.plaphy.2013.03.014]

-
Alam P, Azzam MA, Balawi TA, Raja V, Bhat JA and Ahmad P. (2022). Mitigation of negative effects of chromium(VI) toxicity in faba bean(Vicia faba) plants through the supplementation of kinetin(KN) and gibberellic acid(GA3). Plants. 11:3302. https://www.mdpi.com/2223-7747/11/23/3302, (cited by 2026 Feb 02).
[https://doi.org/10.3390/plants11233302]

-
Arena K, Cacciola F, Dugo L, Dugo P and Mondello L. (2020). Determination of the metabolite content of Brassica juncea cultivars using comprehensive two-dimensional liquid chromatography coupled with a photodiode array and mass spectrometry detection. Molecules. 25:1235. https://www.mdpi.com/1420-3049/25/5/1235, (cited by 2026 Feb 02).
[https://doi.org/10.3390/molecules25051235]

-
Aslam MM, Farhat F, Zulfiqar S, Siddiqui MA, Asim M and Sial MA. (2023). Efficiency of nitrogen, gibberellic acid and potassium on canola production under sub-tropical regions of Pakistan. Scientific Reports. 13:18677. https://www.nature.com/articles/s41598-023-38997-8, (cited by 2026 Feb 12).
[https://doi.org/10.1038/s41598-023-38997-8]

-
Assefa AD, Kim SH, Ko HC, Ro N, Subramanian P, Chung YJ, Lee YH, Hahn BS and Rhee JH. (2023). Leaf mustard (Brassica juncea) germplasm resources showed diverse characteristics in agro-morphological traits and glucosinolate levels. Foods 12:4374. https://www.mdpi.com/2304-8158/12/23/4374, (cited by 2026 Feb 12).
[https://doi.org/10.3390/foods12234374]

-
Carlson TN and Ripley DA. (1997). On the relation between NDVI, fractional vegetation cover, and leaf area index. Remote Sensing of Environment. 62:241-252.
[https://doi.org/10.1016/S0034-4257(97)00104-1]

-
Carmer SG and Swanson MR. (1971). Detection of differences between means: A Monte Carlo study of five pairwise multiple comparison procedures. Agronomy Journal. 63:940-945.
[https://doi.org/10.2134/agronj1971.00021962006300060036x]

-
Castro-Camba R, Sánchez C, Vidal N and Vielba JM. (2022). Plant development and crop yield: the role of gibberellins. Plants. 11:2650. https://www.mdpi.com/2223-7747/11/19/2650, (cited by 2026 Feb 01).
[https://doi.org/10.3390/plants11192650]

-
Davière JM and Achard P. (2013). Gibberellin signaling in plants. Development. 140:1147-1151.
[https://doi.org/10.1242/dev.087650]

-
Dick D and VanderWeide J. (2025). A meta-analysis and systematic review of plant growth regulator use in blueberry production. Frontiers in Plant Science. 16:1632855. https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1632855/full, (cited by 2026 Feb 03).
[https://doi.org/10.3389/fpls.2025.1632855]

-
Dickson A, Leaf AL and Hosner JF. (1960). Quality appraisal of white spruce and white pine seedling stock in nurseries. The Forestry Chronicle. 36:10-13.
[https://doi.org/10.5558/tfc36010-1]

-
Elahi NN, Raza S, Rizwan MS, Albalawi BFA, Ishaq MZ, Ahmed HM, Mehmood S, Imtiaz M, Farooq U, Rashid M and Ditta A. (2022). Foliar application of gibberellin alleviates adverse impacts of drought stress and improves growth, physiological and biochemical attributes of canola(Brassica napus L.). Sustainability. 15:78. https://www.mdpi.com/2071-1050/15/1/78, (cited by 2026 Feb 01).
[https://doi.org/10.3390/su15010078]

-
Fu J, Li L, Wang S, Yu N, Shan H, Shi Z, Li F and Zhong X. (2023). Effect of gibberellic acid on photosynthesis and oxidative stress response in maize under weak light conditions. Frontiers in Plant Science. 14:1128780. https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2023.1128780/full, (cited by 2026 Feb 03).
[https://doi.org/10.3389/fpls.2023.1128780]

-
Gebbers R and Adamchuk, VI. (2010). Precision agriculture and food security. Science. 327:828-831.
[https://doi.org/10.1126/science.1183899]

-
Hauvermale AL, Ariizumi T and Steber CM. (2012). Gibberellin signaling: A theme and variations on DELLA repression. Plant Physiology. 160:83-92.
[https://doi.org/10.1104/pp.112.200956]

-
Hedden P and Thomas SG. (2012). Gibberellin biosynthesis and its regulation. Biochemical Journal. 444:11-25.
[https://doi.org/10.1042/BJ20120245]

-
Heim, KE, Tagliaferro AR and Bobilya DJ. (2002). Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. The Journal of Nutritional Biochemistry. 13:572-584.
[https://doi.org/10.1016/S0955-2863(02)00208-5]

-
Hong J, Lee JH, Kim EA, Shin EJ, Lee S, Joo S, Cha EB, Han H, Lee YC and Nam SY. (2025). Dry-cold stratification combined with pericarp removal: Germination, seedling development, and physiological changes of lilyturf(Liriope muscari(Decne.) LH Bailey). Journal of People, Plants, and Environment. 28:777-798.
[https://doi.org/10.11628/ksppe.2025.28.6.777]

-
Hu Y and Yan Z. (2025). Comprehensive advances in phytochemical components, bioactive functionality, and processing applications of mustard(Brassica juncea (L.) Czern.): A review. Frontiers in Nutrition. 12:1626333. https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2025.1626333/full, (cited by 2026 Feb 12).
[https://doi.org/10.3389/fnut.2025.1626333]

-
Huang H, Wang J, Mao S, Wu Q, Tian Y, Wang F, Wang P, Huang K and Wu Q. (2022). Variation characteristics of glucosinolate contents in leaf mustard(Brassica juncea). Agronomy. 12:2287. https://www.mdpi.com/2073-4395/12/10/2287, (cited by 2026 Feb 02).
[https://doi.org/10.3390/agronomy12102287]

-
Ibrahim MEH, Zhu X, Zhou G, Ali AYA, Elsiddig AMI and Farah GA. (2019). Response of some wheat varieties to gibberellic acid under saline conditions. Agrosystems, Geosciences and Environment. 2:1-7.
[https://doi.org/10.2134/age2019.01.0003]

-
Ibrahim RM, Eltanany BM, Pont L, Benavente F, ElBanna SA and Otify AM. (2023). Unveiling the functional components and antivirulence activity of mustard leaves using an LC-MS/MS, molecular networking, and multivariate data analysis integrated approach. Food Research International. 168:112742.
[https://doi.org/10.1016/j.foodres.2023.112742]

- Iqbal N, Nazar R, Khan MIR, Masood A and Khan NA. (2011). Role of gibberellins in regulation of source-sink relations under optimal and limiting environmental conditions. Current Science. 100:998-1007.
-
Islam MS, Hasan MK, Islam B, Renu NA, Hakim MA, Islam MR, Chowdhury MK, Ueda A, Saneoka H, Raza MA, Fahad S, Barutçular C, Çig F, Erman M and El Sabagh A. (2021). Responses of water and pigments status, dry matter partitioning, seed production, and traits of yield and quality to foliar application of GA3 in mungbean(Vigna radiata L.). Frontiers in Agronomy. 2:596850. https://www.frontiersin.org/journals/agronomy/articles/10.3389/fagro.2020.596850/full, (cited by 2026 Feb 03).
[https://doi.org/10.3389/fagro.2020.596850]

-
Janah LN, Apriliani AAN, Hidayah AN, Adhelia V, Sukirno S and Daryono BS. (2023). Effectiveness of liquid organic fertilizer byproduct of black soldier fly maggot to the growth of mustard plant(Brassica juncea L.). Journal of Tropical Biodiversity and Biotechnology. 8:70530.
[https://doi.org/10.22146/jtbb.70530]

-
Jang IT, Lee JH, Shin EJ and Nam SY. (2023). Evaluation of growth, flowering, and chlorophyll fluorescence responses of Viola cornuta cv. Penny Red Wing according to spectral power distributions. Journal of People, Plants, and Environment. 26:335-349.
[https://doi.org/10.11628/ksppe.2023.26.4.335]

-
Khalil U, Rajwana IA, Razzaq K, Farooq U and Saleem BA. (2023). Physical, biochemical and phytochemical quality variations in grapes treated by exogenous application of gibberellic acid. Erwerbs-Obstbau. 65:2031-2044.
[https://doi.org/10.1007/s10341-023-00897-6]

-
Kim EA, Lee JH and Nam SY. (2024a). Differences in growth characteristics and photochemical responses of Salvia miltiorrhiza Bunge under different LED light qualities in a closed plant factory system. Korean Journal of Medicinal Crop Science. 32:189-201.
[https://doi.org/10.7783/KJMCS.2024.32.4.189]

-
Kim EA, Lee JH, Sunwoo Y, Shin EJ and Nam SY. (2024b). Improvement in growth, external quality, and physiological characteristics of Salvia miltiorrhiza Bunge through photoperiod control. Korean Journal of Medicinal Crop Science. 32:152-162.
[https://doi.org/10.7783/KJMCS.2024.32.3.152]

-
Kim JE, Jung MJ, Jung HA, Woo JJ, Cheigh HS, Chung HY and Choi JS. (2002). A new kaempferol 7-O-triglucoside from the leaves of Brassica juncea L. Archives of Pharmacal Research. 25:621-624.
[https://doi.org/10.1007/BF02976932]

-
Kramer DM, Johnson G, Kiirats O and Edwards GE. (2004). New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynthesis Research. 79:209-218.
[https://doi.org/10.1023/B:PRES.0000015391.99477.0d]

-
Lee GY, Kim YH, Jung JH, Soh HS and Lee SD. (2020). Effect of benzylamino purine(BA) and gibberellic acid(GA3) on growth and flowering in Schlumbergera truncata. Horticultural Science and Technology. 38:419-428.
[https://doi.org/10.7235/HORT.20200040]

- Lee JH. (2023). Effects of light quality, temperature, photoperiod, and GA3 concentration on growth of six Phedimus species. Ph. D. Thesis. Sahmyook University. p.21-49.
-
Lee JH, Kim EA, Sunwoo Y, Cha EB, Joo S, Lee S, Lee JG and Nam SY. (2025b). Light spectrum strategies to optimize yield and physiological performance of Korean thistle(Cirsium setidens(Dunn) Nakai) in a closed-type plant production system. Journal of People, Plants, and Environment. 28:799-823.
[https://doi.org/10.11628/ksppe.2025.28.6.799]

-
Lee JH, Lee S and Nam SY. (2025a). Optimized concentrations of auxinic rooting promoters improve stem cutting propagation efficiency and morphophysiological characteristics in Hedera algeriensis cv. Gloire de Marengo. Horticultural Science and Technology. 43:357-372.
[https://doi.org/10.7235/HORT.20250032]

-
Lee JH and Nam SY. (2024). Growth and photosynthetic responses of Taraxacum coreanum Nakai seedlings according to the shading levels. Horticultural Science and Technology. 42:214-224.
[https://doi.org/10.7235/HORT.20240018]

-
Lee JH, Shin EJ, Kim EA, Jang IT, Lee S and Nam SY. (2024). Effects of different concentrations of exogenous auxins(IAA, IBA, and NAA) on growth and rooting ability of Philodendron hederaceum var. oxycardium(Schott) Croat stem cuttings. Journal of People, Plants, and Environment. 27:279-289.
[https://doi.org/10.11628/ksppe.2024.27.4.279]

-
Lee JH, Shin EJ and Nam SY. (2023). Enhancing propagation of Orostachys spp. offsets through exogenous GA3 application. Horticulturae. 9:1280. https://www.mdpi.com/2311-7524/9/12/1280, (cited by 2026 Feb 02).
[https://doi.org/10.3390/horticulturae9121280]

-
Lin LZ, Sun J, Chen P and Harnly J. (2011). UHPLC-PDA-ESI/HRMS/MSn analysis of anthocyanins, flavonol glycosides, and hydroxycinnamic acid derivatives in red mustard greens (Brassica juncea Coss variety). Journal of Agricultural and Food Chemistry. 59:12059-12072.
[https://doi.org/10.1021/jf202556p]

-
Lovatt CJ. (2013). Properly timing foliar-applied fertilizers increases efficacy: A review and update on timing foliar nutrient applications to citrus and avocado. HortTechnology. 23:536-541.
[https://doi.org/10.21273/HORTTECH.23.5.536]

-
Maršić NK, Mikulič-Petkovšek M, Hudina M, Veberič R and Slatnar A. (2021). Leafy Asian vegetables cultivated on a floating hydroponic system and a substrate culture, during the autumn period in greenhouse. Acta Horticulturae. 1320:413-420.
[https://doi.org/10.17660/ActaHortic.2021.1320.55]

-
Martins AO, Omena-Garcia RP, Oliveira FS, Silva WA, Hajirezaei MR, Vallarino JG, Ribeiro DM, Fernie AR, Nunes-Nesi A and Araújo WL. (2019). Differential root and shoot responses in the metabolism of tomato plants exhibiting reduced levels of gibberellin. Environmental and Experimental Botany. 157:331-343.
[https://doi.org/10.1016/j.envexpbot.2018.10.036]

-
Maxwell K and Johnson GN. (2000). Chlorophyll fluorescence—a practical guide. Journal of Experimental Botany. 51:659-668.
[https://doi.org/10.1093/jexbot/51.345.659]

-
McCabe CP and Burke JI. (2021). Effects of cultivar, seed rate, N rate and plant growth regulator on key processing quality parameters of winter and spring-sown oats. The Journal of Agricultural Science. 159:589-603.
[https://doi.org/10.1017/S0021859621000873]

-
Muniandi SKM, Hossain MA, Abdullah MP and Ab Shukor NA. (2018). Gibberellic acid(GA3) affects growth and development of some selected kenaf(Hibiscus cannabinus L.) cultivars. Industrial Crops and Products. 118:180-187.
[https://doi.org/10.1016/j.indcrop.2018.03.036]

-
Muniz CR, Freire FCO, Viana FMP, Cardoso JE, Sousa CAF, Guedes MIF, van der Schoor R and Jalink H. (2014). Monitoring cashew seedlings during interactions with the fungus Lasiodiplodia theobromae using chlorophyll fluorescence imaging. Photosynthetica. 52:529-537.
[https://doi.org/10.1007/s11099-014-0061-6]

-
Neugart S and Bumke-Vogt C. (2021). Flavonoid glycosides in Brassica species respond to UV-B depending on exposure time and adaptation time. Molecules. 26:494. https://www.mdpi.com/1420-3049/26/2/494, (cited by 2026 Feb 01).
[https://doi.org/10.3390/molecules26020494]

-
Niu J, Liu C, Huang M, Liu K and Yan D. (2021). Effects of foliar fertilization: A review of current status and future perspectives. Journal of Soil Science and Plant Nutrition. 21:104-118.
[https://doi.org/10.1007/s42729-020-00346-3]

-
Noack SR, McBeath TM and McLaughlin MJ. (2010). Potential for foliar phosphorus fertilisation of dryland cereal crops: A review. Crop and Pasture Science. 61:659-669.
[https://doi.org/10.1071/CP10080]

-
Omena-Garcia RP, Martino PB, De Andrade LD, Lima VF, Zsögön A, Ribeiro DM and Nunes-Nesi A. (2025). Gibberellin deficiency uncouples shoot and root growth in tomato and alters morphology and carbon allocation. Planta. 262:137.
[https://doi.org/10.1007/s00425-025-04845-4]

-
Othman YA, Al-Ajlouni MG, A’saf TS, Sawalha HA and Hani MB. (2021). Influence of gibberellic acid on the physiology and flower quality of gerbera and lily cut flowers. International Journal of Agriculture and Natural Resources. 48:21-33. https://ojs.uc.cl/index.php/ijanr/article/view/40615, (cited by 2026 Feb 01).
[https://doi.org/10.7764/ijanr.v48i1.2218]

-
Park CH, Yeo HJ, Park YJ, Morgan AMA, Arasu MV, Al-Dhabi NA and Park SU. (2017). Influence of indole-3-acetic acid and gibberellic acid on phenylpropanoid accumulation in common buckwheat(Fagopyrum esculentum Moench) sprouts. Molecules. 22:374. https://www.mdpi.com/1420-3049/22/3/374, (cited by 2026 Feb 12).
[https://doi.org/10.3390/molecules22030374]

-
Pavlista AD, Santra DK, Schild JA and Hergert GW. (2012). Gibberellic acid sensitivity among common bean cultivars (Phaseolus vulgaris L.). HortScience. 47:637-642.
[https://doi.org/10.21273/HORTSCI.47.5.637]

- Photon Systems Instruments(PSI). (2026). FluorPen FP 110 PAR-FluorPen FP 110 Monitoring Pen MP 100. FluorPen & PAR FluorPen, Photon Systems Instruments. Drásov, Czech Republic. Available via https://handheld.psi.cz/documents/FluorPen_Monitoring_Manual_02_2021.pdf, (cited by 2026 January 03).
-
Prodhan MM, Sarker U, Hoque MA, Biswas MS, Ercisli S, Assouguem A, Ullah R, Almutairi MH, Mohamed HRH and Najda A. (2022). Foliar application of GA3 stimulates seed production in cauliflower. Agronomy. 12:1394. https://www.mdpi.com/2073-4395/12/6/1394, (cited by 2026 Feb 12).
[https://doi.org/10.3390/agronomy12061394]

-
Rademacher W. (2015). Plant growth regulators: backgrounds and uses in plant production. Journal of Plant Growth Regulation. 34:845-872.
[https://doi.org/10.1007/s00344-015-9541-6]

-
Rajala A. (2004). Plant growth regulators to manipulate oat stands. Agricultural and Food Science. 13:186-197.
[https://doi.org/10.2137/1239099041838058]

-
Rezaei EE, Siebert S and Ewert F. (2017). Climate and management interaction cause diverse crop phenology trends. Agricultural and Forest Meteorology. 233:55-70.
[https://doi.org/10.1016/j.agrformet.2016.11.003]

-
Robil JM, Awale P, McSteen P and Best NB. (2025). Gibberellins: Extending the green revolution. Journal of Experimental Botany. 76:1837-1853.
[https://doi.org/10.1093/jxb/erae476]

-
Sari Ö. (2024). Analyzing the effects of different GA3 applications on plant root architecture and above-ground properties in tulip cultivars.. Horticultural Science(Prague). 51:244-254.
[https://doi.org/10.17221/174/2023-HORTSCI]

- Shaddad MAK, Abd El-Samad HM and Mostafa D. (2013). Role of gibberellic acid(GA3) in improving salt stress tolerance of two wheat cultivars. International Journal of Plant Physiology and Biochemistry. 5:50-57. https://academicjournals.org/journal/IJPPB/article-abstract/CF7523041977, (cited by 2026 Feb 01).
-
Shahi S, Yadav RK, Kushwaha SP, Singh M and Singh P. (2022). Effect of plant growth regulators on biochemical studies of mustard[Brassica juncea(L.) Czern & Coss.] under sodic soil. International Journal of Plant and Soil Science. 34:280-286. https://journalijpss.com/index.php/IJPSS/article/view/1908, (cited by 2026 Feb 12).
[https://doi.org/10.9734/ijpss/2022/v34i1931114]

-
Shin EJ, Lee JH and Nam SY. (2024). Evaluation of growth, vegetation indices, and photosynthesis of Cichorium intybus L. seedlings as affected by LED light qualities in a closed nursery facility. Horticultural Science and Technology. 42:350-364.
[https://doi.org/10.7235/HORT.20240029]

-
Stirbet A and Govindjee G. (2011). On the relation between the Kautsky effect(chlorophyll a fluorescence induction) and photosystem II: Basics and applications of the OJIP fluorescence transient. Journal of Photochemistry and Photobiology B: Biology. 104:236-257.
[https://doi.org/10.1016/j.jphotobiol.2010.12.010]

-
Sun H, Cui H, Zhang J, Kang J, Wang Z, Li M, Yi F, Yang Q and Long, R. (2021). Gibberellins inhibit flavonoid biosynthesis and promote nitrogen metabolism in Medicago truncatula. International Journal of Molecular Sciences. 22:9291. https://www.mdpi.com/1422-0067/22/17/9291, (cited by 2026 Feb 12).
[https://doi.org/10.3390/ijms22179291]

-
Tian Y and Deng F. (2020). Phytochemistry and biological activity of mustard(Brassica juncea): A review. Cyta-Journal of Food. 18:704-718.
[https://doi.org/10.1080/19476337.2020.1833988]

-
Tomer E. (1984). Inhibition of flowering in mango by gibberellic acid. Scientia Horticulturae. 24:299-303.
[https://doi.org/10.1016/0304-4238(84)90114-6]

- Vinu V, Singh N, Vasudev S, Yadava DK, Kumar S, Naresh S, Bhat SR and Prabhu KV. (2013). Assessment of genetic diversity in Brassica juncea(Brassicaceae) genotypes using phenotypic differences and SSR markers. Revista de Biología Tropical. 61:1919-1934.
-
Wenzel CL, Williamson RE and Wasteneys GO. (2000). Gibberellin-induced changes in growth anisotropy precede gibberellin-dependent changes in cortical microtubule orientation in developing epidermal cells of barley leaves. Kinematic and cytological studies on a gibberellin-responsive dwarf mutant, M489. Plant Physiology. 124:813-822.
[https://doi.org/10.1104/pp.124.2.813]

-
Wu X, Gong D, Zhao K, Chen D, Dong Y, Gao Y, Wang Q and Hao GF. (2024). Research and development trends in plant growth regulators. Advanced Agrochem. 3:99-106. https://www.sciencedirect.com/science/article/pii/S2773237123000989?via%3Dihub, (cited by 2026 Feb 01).
[https://doi.org/10.1016/j.aac.2023.11.005]

-
Xue J and Su B. (2017). Significant remote sensing vegetation indices: A review of developments and applications. Journal of Sensors. 2017:1353691.
[https://doi.org/10.1155/2017/1353691]

-
Yamaguchi S. (2008). Gibberellin metabolism and its regulation. Annual Review of Plant Biology. 59:225-251.
[https://doi.org/10.1146/annurev.arplant.59.032607.092804]

-
Yan G, Yu P, Tian X, Guo L, Tu J, Shen J, Yi B, Fu T, Wen J, Liu K, Ma C and Dai C. (2021). DELLA proteins BnaA6.RGA and BnaC7.RGA negatively regulate fatty acid biosynthesis by interacting with BnaLEC1s in Brassica napus. Plant Biotechnology Journal. 19:2011-2026.
[https://doi.org/10.1111/pbi.13628]

- Zettl A. (2026). Converting Colors. Converting Colors website. Available via https://convertingcolors.com, (cited by 2026 January 03).
-
Zhang D, Yu H, Gao L, Wang J, Dong H, Guo Y and Hu S. (2023). Genetic diversity in oilseed and vegetable mustard (Brassica juncea L.) accessions revealed by nuclear and mitochondrial molecular markers. Agronomy. 13:919. https://www.mdpi.com/2073-4395/13/3/919, (cited by 2026 Feb 01).
[https://doi.org/10.3390/agronomy13030919]

-
Zhang W, Cheng X, Jing Z, Cao Y, Yuan S, Zhang H and Zhang Y. (2024). Exogenous GA3 enhances nitrogen uptake and metabolism under low nitrate conditions in ‘Duli’(Pyrus betulifolia Bunge) seedlings. International Journal of Molecular Sciences. 25:7967. https://www.mdpi.com/1422-0067/25/14/7967, (cited by 2026 Feb 01).
[https://doi.org/10.3390/ijms25147967]