Attenuating Effect of Geijigadaehwang-tang in Aggression Animal Model by Isolation Rearing Based on Experimental Study and Network Pharmacology
This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Social isolation reportedly induces excessive aggression and impairs hippocampal neurogenesis. In this study, we investigated whether Geijigadaehwang-tang (GDT), a traditional herbal formulation containing Rhei Radix et Rhizoma and Cinnamomi Ramulus, could ameliorate such behavioral and neurogenic deficits.
We assessed aggressive behaviors in socially isolated mice. Isolation markedly increased aggressive responses compared to sham controls. GDT treatment significantly reduced the number of attacks on days 3 and 7 and lowered both the attack duration and biting frequency. Immunohistochemical analysis of the dentate gyrus revealed that the isolation substantially reduced DCX-positive immature neuron numbers, whereas GDT administration partially restored these counts. Network pharmacology profiling has identified numerous GDT-associated compounds that interact with neuroendocrine and neurotransmission-related targets, including the dopaminergic, adrenergic, GABAergic, and CRH receptor pathways. Enrichment analysis highlighted neuroactive ligand-receptor interaction, PI3K-Akt signaling, calcium signaling, apoptosis regulation, and steroid hormone biosynthesis as major functional clusters, supporting the role of GDT in neural plasticity and stress-related behavior.
GDT attenuated isolation-induced aggression and partially rescued impaired hippocampal neurogenesis. Integrated network analysis suggests that its multimodal actions might involve the modulation of neurotransmitter systems, neuroendocrine pathways, and pro-survival signaling cascades. GDT represents a promising multitarget therapeutic candidate for stress-related behavioral dysregulation and neurogenic impairment.
Keywords:
Neurodegenerative Disorders, Inflammation, Isolation, Aggression, Geijigadaehwang-tang, Network PharmacologyINTRODUCTION
Pathological aggression is associated with a variety of psychiatric disorders and disease states including brain injury, seizure and dementia disorders, schizophrenia, depression, personality disorders, and post-traumatic stress disorder. Long-term individual housing of mice during young adulthood increases aggressive behavior against conspecifics; this phenomenon has been termed “isolation-induced aggression” (Yen, 1959; Anton et al., 1968; Valzelli, 1969; Brain and Nowell, 1971; Valzelli, 1973; Miczek and O'Donnell, 1978). Isolation-induced aggression is the one of the most frequently used method to induce aggressive behavior in mice. Apart from attacking behavior, which includes lateral threat, aggression, and tail rattle, other behavioral elements including social interaction, exploration, avoidance, defense, inactivity, and self-care are also observed (Olivier and Young, 2002). The involvement of different neurotransmitters in mediating aggressive behavior has been studied extensively. Serotonin (5-HT) plays an important role, but noradrenaline (NA), dopamine (DA), and GABA are also involved (Eichelman, 1990).
Geijigadaehwang-tang (GDT) was first described in the ancient medical text Shanghanlun (the late Eastern Han dynasty, China), and this remedy has since been widely used to treat enteritis (Ahn et al., 2020). From the perspective of formula composition, GDT is based on Gyejigajakyak-tang, which consists of Cinnamomi Ramulus, Paeoniae Radix, Zingiberis Rhizoma Recens, Jujubae Fructus, and Glycyrrhizae Radix, with the addition of Rhei Radix et Rhizoma (Rhubarb) (The Association of Herbal Formula Textbook Publication, 2023). Previous studies have shown antioxidant and neuroprotective effects of Cinnamomi Ramulus (Ulbricht et al., 2011), anti-inflammatory, antioxidant, and anti-Alzheimer's activities of ginger (Seibel et al., 2021), and neuroprotective and antineurodegenerative effects of peony (Du et al., 2020). Collectively, these findings suggest that GDT has potential as a therapeutic agent for neuropathological conditions.
Schizophrenia is a mental illness characterized by severe cognitive and emotional dysfunction. Aggressive behavior is reported to be a key clinical feature of the disorder (Hodgins et al., 2017; Li et al., 2020). The social environment, particularly during early development and adolescence, plays a crucial role in shaping brain function. In humans, social isolation is a major environmental risk factor for the development of schizophrenia (Fone and Porkes, 2008). Against this backdrop, the rodent social isolation model is widely used as a valid animal model for studying the pathophysiology of schizophrenia. This model effectively replicates core symptoms observed in patients with schizophrenia, such as social withdrawal, sensorimotor gating deficits, and abnormally increased aggression (Fone and Porkes, 2008; Li et al., 2017). Therefore, evaluating drug efficacy using a social isolation-induced aggression model provides important academic evidence for elucidating the therapeutic mechanisms of schizophrenia-related mental disorders.
In herbology, Rhei Radix et Rhizoma (Daehwang) is traditionally used to treat constipation, abdominal pain, diarrhea, jaundice, epistaxis, conjunctivitis, sore throat, appendicitis, bruising, amenorrhea, and burns (Herbology Textbook Committee, 2020). Classical formulas containing Rhei Radix et Rhizoma, such as Daeseunggi-tang, Dohaekseunggi-tang, Jeodang-tang, and Sasim-tang, were also prescribed for patients showing symptoms resembling modern psychiatric disorders, including hallucinations, mania, agitation, and aggressive behavior (Ahn et al., 2020). Rhubarb contains anthraquinone derivatives, such as emodin, aloe-emodin, chrysophanol, physcion, and sennosides, which exhibit neuroprotective properties (Li et al., 2019); these characteristics suggest that rhubarb may exert beneficial effects on pathological aggression.
Recent in vivo and in silico studies have reported that rhubarb attenuates seizures and hippocampal neurodegeneration in trimethyltin-induced neurotoxicity models (Choi et al., 2024). Moreover, GDT has been shown -both in vitro and in vivo- to suppress trimethyltin-induced seizures and hippocampal injury, likely through modulation of inflammatory and oxidative stress pathways (Lee et al., 2022). Based on these findings, the present study investigated the effects of GDT on abnormal aggression induced by prolonged social isolation and on hippocampal neurogenesis.
In this study, the therapeutic potential of GDT for isolation-induced aggression in mice was evaluated by integrating experimental research with network pharmacology.
MATERIALS AND METHODS
1. Herbal medicine extraction
Cinnamomi Ramulus, Paeoniae Radix Alba, Zingiberis Rhizoma Recens, Zizyphi Fructus, Glycyrrhizae Radix et Rhizoma, and Rhei Radix et Rhizoma were purchased from Omniherb (Daegu, Republic of Korea), Nanum Co., Ltd.(Yeoncheon, Republic of Korea), or Bonchowon (Yeoncheon, Republic of Korea). GDT (a total of 141 g, Table 1) was prepared by a 150-min cooling reflux extraction using 1.5 L of distilled water as a solvent (100±2°C) after maceration at 4°C for 2 hours. The extract was filtered through a filter paper (6 mm, No. 1, Advantec MFS Inc., Tokyo, Japan), concentrated using a vacuum evaporator (N-1000; EYELA, Bohemia, NY, USA) at 70°C, and lyophilized using a freeze dryer (FD8512; Ilshin Lab Co. Ltd., Daejeon, Republic of Korea). GDT was prepared three times with a yield of 12.3±0.7% (12.0±0.7 g). GDT powder was stored in a freezer at -20°C. The diluted solution was filtered using a 20-Ø filter (Minisart® High Flow; DAWIMbio, Hanam-si, Korea) for cell treatment. The final concentration used for treatment was 5% or less to prevent osmotic shock.
2. Animal experiments
Six-week-old male C57BL/6 mice were sourced from Central Lab Animal Inc. (Seoul, Korea) and given a week to acclimatize. Following Olivier's suggestion and Ma X's study, we selected C57BL6 male mice to induce offensive behavior via social isolation. The animals were housed under the following controlled environmental conditions: 23±2°C temperature, 50±5% relative humidity, and a 12-h artificial light cycle (08:00-20:00). A standard diet was provided to the mice. All experimental procedures were conducted in accordance with the protocols approved by the Institutional Animal Care and Use Committee at Dongshin University (approval no.: DSU 2023-04-03) and Chonnam National University (approval no.: CNU2024-112).
3. Isolation and GDT extract administration
Mice were acclimated to stable environmental conditions for one week prior to the start of the study. Animals were divided into three groups: control, isolation + vehicle treatment, and isolation + GDT treatment. To induce aggression, mice in the isolation groups were singly housed in individual cages (254 × 145 × 127 mm) for eight weeks with ad libitum access to food and water. This is a condition known to enhance aggressive behavior in mice [isolation-induced aggression, (Olivier and Young, 2002)]. In contrast, control mice were socially reared in groups of 5-6 per cage (200 × 260 × 130 mm).
To facilitate the manifestation of aggressive behavior, 6-week-old male C57BL/6 mice were purchased one week prior to the behavioral tests and acclimated to serve as smaller-sized opponents in the neutral arena. These group-housed intruders were utilized because they reliably provoke attacks from isolated residents without initiating the aggression themselves.
To evaluate the inhibitory efficacy of GDT on isolationi-nduced aggression, isolated mice were subdivided into the isolation+GDT and isolation+vehicle groups after an initial encounter in the neutral arena. The isolation + GDT group was administered GDT at 180 mg/kg/day, administered twice daily for 7 days, and the isolation + vehicle group was administered the same amount of water twice daily. The GDT dosage was determined based on previous findings regarding its neuroprotective efficacy. Aggressive behavior was assessed at baseline (Day 0, prior to the initiation of drug administration) and subsequently on the 3rd and 7th days of the treatment period.
4. Aggressive behavior assessment in a neutral arena
Aggressive behavior was evaluated using a neutral arena paradigm. As noted by Olivier and Young (2002), neutral environments are particularly effective for observing a clear mixture of offensive and defensive behaviors that may occur less frequently during home cage confrontations. Isolated residents (large) and socially reared opponents (small) were placed together in a novel cage (260 × 420 × 180 mm) for a 1:1 encounter.
To minimize external interference, the experimenter left the room during the session, and the interactions were recorded using a Quickcam system (Logitech, Lausanne). Aggression was quantified during the first 15 minutes of the encounter by measuring the frequency and total duration of fighting episodes, specifically focusing on biting behavior. All behavioral analyses were conducted by observers who were blinded to the housing and treatment conditions of the animals.
5. Cotton bud biting test
Aggressive behavior was assessed using the cotton bud biting test, following the procedure described previously (Park et al., 2015). Each mouse was gently held by the experimenter while a sterilized cotton bud was presented near its face. A bite directed toward the cotton bud was recorded as an aggressive response. Each animal underwent 10 trials, and the total number of biting events was used for analysis.
6. Immunohistochemical analysis
Immunohistochemistry was conducted according to the protocol described in our previous study (Lee et al., 2022). Fixed brain hemispheres were longitudinally sectioned at a thickness of 4 μm. The sections were subjected to immunolabeling using rabbit anti-DCX (1:500; Cell Signaling Technology, Beverly, MA, USA) primary antibodies. After incubation, the sections were treated with a biotinylated goat anti-rabbit IgG secondary antibody (Vector ABC Elite Kit; Vector Laboratories, Burlingame, CA, USA), followed by an avidin-biotin complex solution from the same kit. Signal detection was performed using a diaminobenzidine (DAB) chromogenic substrate (SK-4100; Vector Laboratories). The slides were then counterstained with hematoxylin and mounted. Digital images of the stained sections were obtained using the Motic EasyScan Digital Slide Scanner (Motic, Hong Kong, China), and staining intensity in the hippocampus was quantified using ImageJ software (NIH, Bethesda, MD, USA).
7. Network pharmacology analysis
The public database, Traditional Chinese Medicine Systems Pharmacology (TCMSP; https://tcmsp-e.com/tcmsp.php, version 2.3), was searched for small molecules of Cinnamomi Ramulus and Rhei Radix et Rhizoma. The information and structures of small molecules were confirmed using the ChEMBL database (https://www.ebi.ac.uk/chembl/, ChEMBL 32 version), PubChem (https://pubchem.ncbi.nlm.nih.gov/, accessed on 2, May 2023), ChemSpider (https://www.chemspider.com/, version 2022.0.66.0), and ChemDraw software 22.2.0.3300 (PerkinElmer Informatics, Revvity Signals Software, Inc.). All small molecules were screened through in silico integrative ADME model with oral bioavailability (OB) ≥ 30% and drug likeness (DL) ≥ 0.18 (Lee et al., 2018; Qi et al., 2020). OB refers to the relative amount of the drugs absorbed by the body into the systemic blood circulation and rate after oral administration, and DL refers to the likeness index of the molecules and the known marketed drugs (Chen et al., 2021).
Genes related to 25 active components selected through the aforementioned ADME screening were collected from the SwissTargetPrediction databases (http://www.swisstargetprediction.ch/, accessed in 4, May 2023) with 'Homo sapiens' as the species and probability ≥ 0 and STITCH (http://stitch.embl.de/, version 5.0) with 'Homo sapiens' as the species and medium confidence (≥ 0.400). In this database, we used a probability derived from our cross-validation analysis to rank the targets and estimate the accuracy of the predictions (Gfeller et al., 2014). Potential target genes were identified in GeneCards: Human Gene Database (https://www.genecards.org/; version 5.15). Potential target genes were selected as the intersection of the genes related to active small molecules and schizophrenia-related genes.
A PPI network and cluster analysis was performed using potential target genes in the STRING database (https://string-db.org/, version 11.5) with a medium confidence score (≥ 0.400). PPI networks were analyzed by identifying sub-networks or modules that exhibited specific topological and/or functional characteristics (Vella et al., 2018). The topological features of the related PPI network were used to identify possible combinations of hit targets (Mukherjee et al., 2021). Sophisticated network-based tools have been found to predict potential disease genes (Pak et al., 2022). PPI analyses were conducted with the STRING database (https://string-db.org/, version 11.5) with a medium confidence score (≥ 0.400), and the topology of PPI networks was evaluated using Cytoscape version 3.8.2 (https://cytoscape.org/) (Pak et al., 2022). Cluster analysis was performed using K-means, and after dividing into a total of five clusters, GO analysis was performed with Benjamini-Hochberg (p < 0.05).
Signal pathway analysis was performed using the DAVID informatics Resources (https://david.ncifcrf.gov; DAVID 2021 version) and KEGG: Kyoto Encyclopedia of Genes and Genomes (https://www.genome.jp/kegg/) with p < 0.05. The network was visualized using Cytoscape version 3.8.2.
8. Statistical Analysis
All data are presented as the mean±standard error (SE). Statistical significance was assessed using an unpaired t-test with GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA). Statistical significance was set at p < 0.05.
RESULTS
1. Anti-aggressive activity of GDT against isolation induced aggression
To determine the anti-aggressive potential of GDT, isolated mice were treated with GDT (180 mg/kg/day). Across the experimental period, mice treated with GDT displayed a marked reduction in aggressive biting behavior compared with the water-treated controls. On Day 0, no significant difference was observed between groups. By Day 3, the isolation + GDT group showed a significant decrease in the number of biting attacks relative to the isolation+vehicle group (p < 0.05). This suppressive effect on aggression was further strengthened by Day 7, where GDT-treated mice exhibited an even greater reduction in attack frequency (p < 0.01) (Fig. 1A).
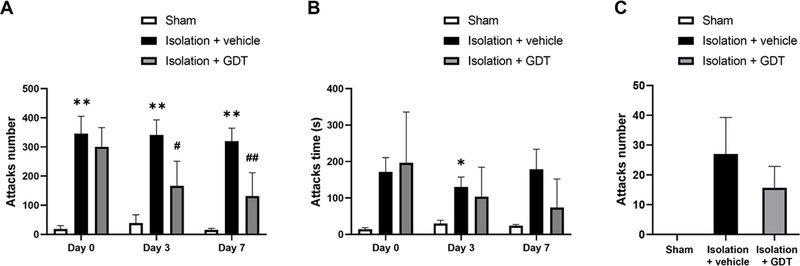
Effects of GDT on isolation-induced aggressive behavior in mice.(A) Number of attacks measured on Days 0, 3, and 7. Social isolation significantly increased attack number, whereas GDT treatment markedly reduced aggressive responses on Days 3 and 7. (B) Duration of attacks across the same time points. GDT-treated mice showed shorter attack times compared with isolated controls. (C) Cotton Biting frequency assessed in sham, isolation + vehicle, and isolation + GDT groups. GDT administration decreased biting frequency relative to isolated controls. Data are presented as mean ± SE. * p < 0.05, ** p < 0.01 vs. Sham; # p < 0.05, ## p < 0.01 vs. Isolation + vehicle.
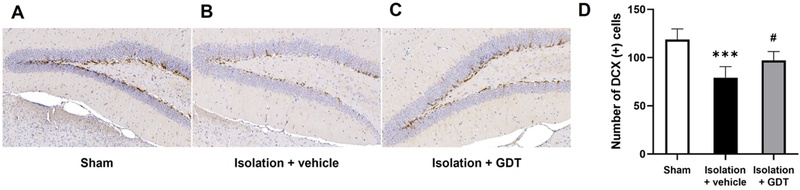
Effects of GDT on hippocampal neurogenesis in socially isolated mice.(A–C) Representative immunohistochemical staining of DCX-positive immature neurons in the dentate gyrus of the hippocampus in the Sham (A), Isolation + vehicle (B), and Isolation + GDT (C) groups. Social isolation markedly reduced the number of DCX-positive cells, whereas GDT treatment partially restored DCX expression. (D) Quantification of DCX-positive cells among groups. Isolation significantly decreased DCX-positive cell counts compared with sham controls, while GDT administration significantly increased DCX-positive cells relative to the isolated vehicle group. Data are presented as mean ± SE. *** p < 0.001 vs. Sham; # p < 0.05 vs. Isolation + vehicle.
Attack duration showed no significant difference between groups at baseline (Day 0). By Day 3, the GDT-treated mice exhibited a noticeable reduction in total attack time compared with the vehicle-treated mice; however, the difference did not reach statistical significance. This decreasing trend continued through Day 7, where GDT-treated animals maintained shorter attack durations than vehicle-treated animals. Overall, GDT administration was associated with a gradual attenuation of aggressive attack time across the observation period (Fig. 1B).
Isolation markedly increased aggressive biting behavior in the isolation groups compared with the control group, as reflected by the elevated number of attacks. Administration of GDT to isolated mice reduced the biting frequency, resulting in substantially fewer aggressive responses than among vehicle-treated mice. Although variability remained high, the overall pattern indicates that GDT attenuates isolation-induced aggression (Fig. 1C).
2. Inhibitory efficacy of GDT against isolation-induced neurodegeneration
In the hippocampus, social isolation led to a reduction in the number of DCX-positive immature neurons compared with the sham group, indicating impaired neurogenesis following isolation-induced aggressive behavior. GDT administration partially restored DCX expression in isolated mice, showing an increased number of DCX-positive cells relative to the untreated isolation group. Although not fully recovered to sham levels, the upward trend suggests that GDT mitigates the isolation-associated decline in hippocampal neurogenesis.
3. Network pharmacology analysis
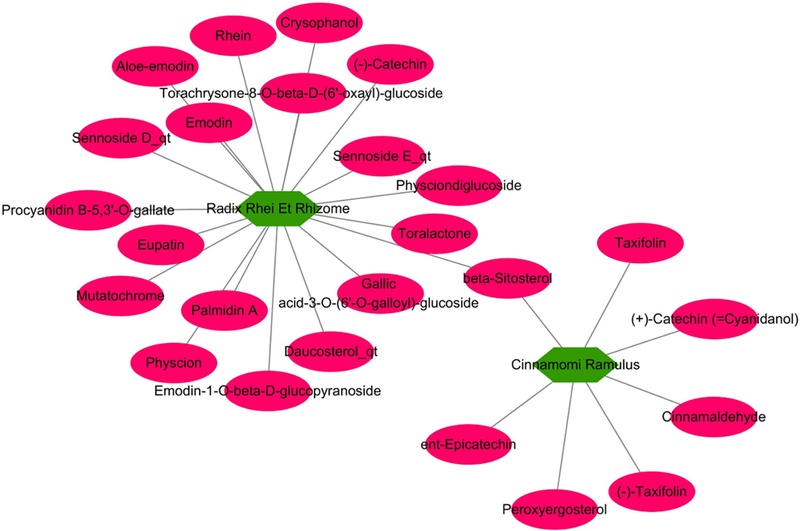
TCMSP was searched, and a total of 297 small molecules were identified; of these, 211 and 90 small molecules were related to Cinnamomi Ramulus and Rhei Radix et Rhizoma, respectively, and 4 molecules were overlapping. Then, there were a total of 21 active small molecules that satisfied the criteria of OB ≥ 30 and DL ≥ 0.18 (Lee et al., 2018), and 4 active small molecules were set the indicator molecules for Cinnamomi Ramulus and Rhei Radix et Rhizoma. The 23 active molecules were grouped as 10 anthraquinones (aloe-emodin, chrysophanol, emodin, emodin 1-O-beta-D-glucopyranoside, palmidin A, physcion, physcion diglucoside, rhein, sennoside D_qt, and sennoside E_qt), 3 steroids (beta-sitosterol, daucosterol_qt, and peroxyergosterol), 3 phenols (cinnamaldehyde, gallic acid-3-O-(6'-O-galloyl)-glucoside, and torachrysone-8-O-beta-D-(6'-oxayl)-glucoside), 5 flavonoids (catechin, epicatechin, eupatin, procyanidin B-5,3'-gallate, and taxifolin), 1 naphthopyran (toralactone), and 1 carotenoid (mutatochrome) (Fig. 3).
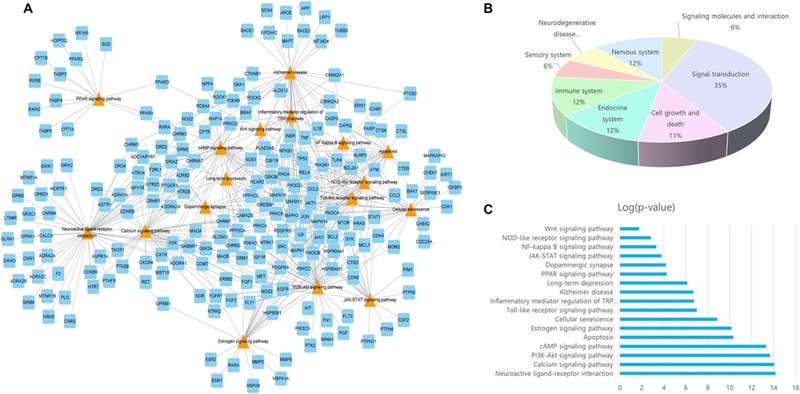
As a result of a gene search related to 23 active molecules from the STITCH and SwissTargetPrediction databases, a total of 813 genes were retrieved (Fig. 3). Although (+)-catechin and (-)-catechin, taxifolin and (-)-taxifolin are enantiomers, the same genes were found in the aforementioned two DBs without distinction. In the GeneCards DB, a total of 14,037 genes related to schizophrenia were identified, and a total of 480 genes overlapped with 1,265 genes related to the small molecules. This network consisted of 457 nodes and 8,122 edges (Fig. 4A). According to the results of the PPI analysis (Fig. 4B), the genes with the top 10 of closeness centrality and node were AKT1, ALB, TNF, TP53, IL6, CTNNB1, SRC, EGFR, MAPK3, and VEGFA. There was also a high value of betweenness centrality nodes for AKT1, ALB, TP53, TNF, ESR1, IL6, CTNNB1, SRC, PPARG, and EGFR. The degree of a node is the number of edges connected to it (Lee et al., 2020). A node with a high degree of betweenness centrality and closeness centrality values plays a highly important role in the network (Zhang et al., 2020). Some proteins in biological activities have a close relationship and have the same or similar functions, which can be defined as a cluster (Hu et al., 2019). Fourteen clusters were obtained using the MCODE plugin cluster analysis of the PPI network of schizophrenia (Fig. 5A). Among the 14 clusters, the 6th cluster was found to be most related to schizophrenia, in which the related genes were ADRA2A, DRD4, SLC6A2, HSD17B1, GABBR1, ADRA2C, OXTR, HSD11B1, SRD5A1, AVPR1A, SLC6A3, STS, GRK6, ADRBK2, and CYP17A1.
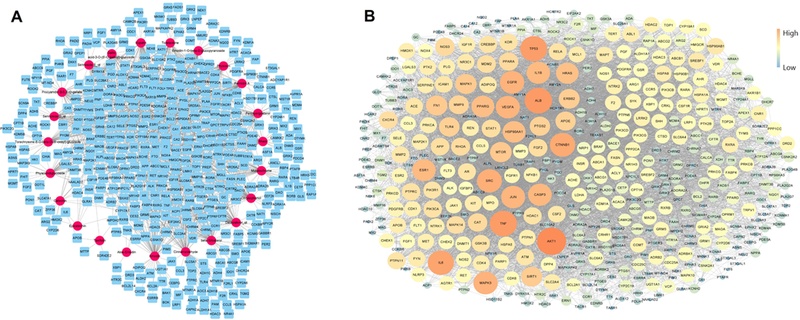
Network analysis of active small molecules and potential target genes related to Schizophrenia disease.(A) 480 nodes and 1,265 edges; pink ovals are active small molecules and cyan rectangles are potential target genes. (B) Protein-protein interactions (PPIs); Core gene is the larger size of the circle and the more orange color.
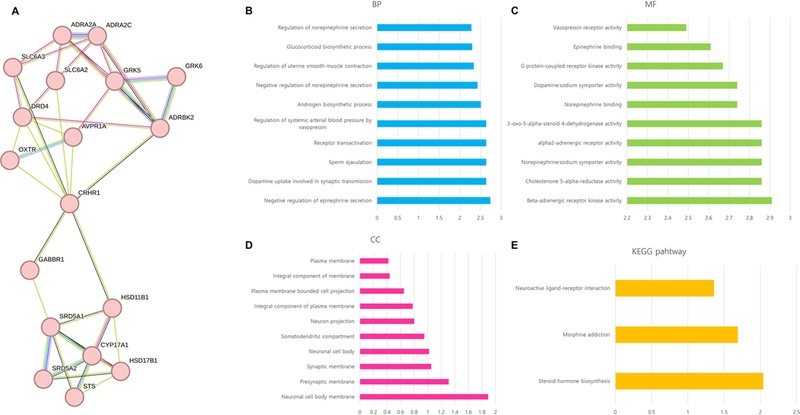
Cluster of Schizophrenia disease PPI network and Gene ontology analysis.the genes included in the cluster most related to schizophrenia disease (A), biological process (B), molecular function (C), Cellular component (D), and KEGG pathway (E).
Analyzing these selected genes in the String DB formed a network with 18 nodes and 40 edges, and gene ontology (GO) including biological process (BP), molecular function (MF), and cellular component (CC) analysis showed only the top 10 terms (Fig. 5B-D). In the KEGG pathway, steroid hormone biosynthesis, morphine addiction, and neuroactive ligand-receptor interaction (Fig. 5E). Diseases related to these features include oppositional defiant disorder, attention deficit hyperactivity disorder, endogenous depression, impulse control disorder, steroid inherited metabolic disorder, drug dependence, cognitive disorder, and diseases of mental health, and tissue expression acts on the dopaminergic system.
The signaling pathways and functions of genes were analyzed using the DAVID and KEGG databases with p < 0.05 (Xu et al., 2022). A total of 17 pathways were related to schizophrenia. The pathways were as follows: neuroactive ligand-receptor interaction, calcium signaling pathway, PI3K-Akt signaling pathway, cAMP signaling pathway, apoptosis, estrogen signaling pathway, cellular senescence, Toll-like receptor signaling pathway, inflammatory mediator regulation, TRP channels, Alzheimer disease, long-term depression, PPAR signaling pathway, dopaminergic synapse, JAK-STAT signaling pathway, NF-kappa B signaling pathway, NOD-like receptor signaling pathway, and Wnt signaling pathway. As a result of the network analysis between pathways and genes, 17 pathways related to schizophrenia were related to 174 genes. Among them, the neuroactive ligand-receptor interaction pathway involved 57 genes from this network. Analyzing the KEGG categories, signal transduction accounted for 35.3%, followed by cell growth and death, the endocrine system, the immune system, and the nervous system with 11.8% each (Fig. 6).
DISCUSSION
Social isolation is a well-established stressor that induces behavioral and neurobiological alterations including heightened aggression and impaired hippocampal neurogenesis (Olivier and Young, 2002). In the present study, isolated mice showed a clear increase in aggressive biting behavior together with a reduction in DCX-positive immature neurons in the dentate gyrus. These findings are consistent with those of previous reports demonstrating that chronic social isolation increases stress-related aggression (Ibi et al., 2008) and suppresses hippocampal neurogenesis through glucocorticoid-mediated and inflammation-mediated mechanisms (Stranahan et al., 2006).
The following herbs or constituents of GDT have been reported to have neuroprotective and behavioral abnormality control effects. Ethanol extracts of Cinnamomi Ramulus showed anti-inflammatory activity in LPS-treated microglial BV2 cells and showed neuroprotective effects on tunicamycin-treated neuroblastoma SH-SY5Y cells (Hwang et al., 2009; Yang et al., 2017; Liu et al., 2018). Paeoniflorin, one of the bioactive compounds of Paeoniae Radix, was reviewed for its antidepressive activity via increasing monoamine neurotransmitter levels, inhibiting the hypothalamic-pituitary-adrenal axis, and elevating brain derived neurotrophic factor levels (Guo et al., 2023). Zingiberis Rhizoma Recens has been recognized for its great potential in treating neurological diseases such as multiple sclerosis, Parkinson's disease, Alzheimer's disease, migraine, and epilepsy due to its antioxidant, immunomodulatory, and anti-inflammatory properties (Arcusa et al., 2022). Glycyrrhizae Radix et Rhizoma has been systematically reviewed for its antioxidant, anti-inflammatory, neuroprotective, antidepressant, and sedative effects (Pastorino et al., 2018). An ethanol extract of Fructus Jujubae has been shown to reduce locomotor activity, relax muscles, and have anti-anxiety effects (Upadhyay et al., 2022). Rhei Radix et Rhizoma acts on the central nervous system to exhibit antipyretic effects (Cao et al., 2017), and its main ingredient, chrysophanol, protects the brain in cerebral ischemia-reperfusion in mice and improves learning and memory (Li and Jiang, 2018). Therefore, GDT has the potential to act on the central nervous system to induce behavioral changes such as alleviation of aggression.
Although GDT has traditionally been utilized for gastrointestinal disorders, recent clinical reports suggest its significant potential in modulating central nervous system and neuropsychiatric conditions. Previous studies have documented a case where GDT administration markedly reduced pain intensity and neuropathic pain scores in a patient suffering from central post-stroke pain (Park, 2020). Furthermore, GDT has demonstrated efficacy in alleviating anxiety and somatic symptoms in patients with somatic symptom disorder through multi-center case series (Yun et al., 2021), and has been reported to improve sleep quality in patients with insomnia disorder (Doo and Lee; 2024). Its clinical utility extends to various psychiatric manifestations, including panic disorder, obsessive-compulsive behavior, and delusions, where GDT-based diagnostic approaches for Greater Yin disease yielded favorable clinical outcomes (Ha et al., 2020). These clinical findings support the hypothesis that GDT possesses the potential to modulate CNS plasticity and emotional regulation systems, consistent with the neurogenic and anti-aggressive effects observed in the present study. Nevertheless, further molecular biological investigations are warranted to fully elucidate the underlying mechanisms supporting these clinical observations.
Administration of GDT attenuated both behavioral and cellular consequences of isolation. GDT markedly reduced aggressive attacks and partially restored the number of DCX-positive cells. The improvement in neurogenesis is particularly notable, as DCX expression reflects the survival and maturation of newly generated granule cells. Restoration of this population suggests that GDT may counteract isolation-related deficits by modulating neuroinflammatory signaling, oxidative stress, or stress-hormone pathways-mechanisms known to regulate adult hippocampal neurogenesis (Huang et al., 2015; Ryan and Nolan, 2016; Toda et al., 2019)
Aggression and neurogenesis are closely linked as follows. Socially isolated female Wistar albino rats had significantly lower total neuronal counts and DCX-positive neuroblast counts in the dentate gyrus of the hippocampus compared to non-isolated rats (Yavaş and Ersoy, 2025). Aggressive foxes have suppressed hippocampal neurogenesis and significantly higher cortisol levels compared to docile foxes (Alexandrovich et al., 2023). Glucocorticoids secreted by stress inhibit neuronal regeneration in the hippocampus, which is known to be involved in anxiety and depression (Snyder et al., 2011). These studies suggest that the impairment of neurogenesis in the hippocampus plays a pivotal role in the manifestation of aggressive behavior.
Although DCX recovery in the GDT-treated group did not reach sham levels, the partial restoration indicates a biologically meaningful effect. Subtle increases in neurogenesis have been shown to improve behavioral outcomes in stress-based models (Snyder et al., 2011). Therefore, modest neurogenic enhancement by GDT could still confer substantial functional benefits.
Network pharmacology analysis provided additional mechanistic insight into the multi-target actions of GDT. GDT contains numerous bioactive compounds-such as emodin, aloe-emodin, rhein, palmidin A, taxifolin, catechins, and cinnamaldehyde-that have documented neuroprotective, anti-inflammatory, and neuromodulatory effects (Lee et al., 2022; Choi et al., 2024).
Emodin is plentiful in three plant families: Polygonaceae (Rheum, Rumex, and Polygonum spp.), Fabaceae (Cassia spp.), and Rhamnaceae (Rhamnus, Frangula, and Ventilago spp.) (Mitra et al., 2022). Mizuno et al. reported that emodin could be a new class of prodrugs for antipsychotics by attenuating EGF receptor signaling and improving behavioral deficits (Mizuno et al., 2008). Emodin also attenuated methamphetamine-induced increases in dopamine and its metabolites and decreases in serotonin and its metabolites (Mizuno et al., 2010). Along with the bright spots in several studies, emodin has the potential to affect various signaling pathways and be stimulating (Mitra et al., 2022). Dietrich-Muszalska et al. reported that epicatechin, a major polyphenol component of green tea, significantly reduced haloperidol-induced plasma lipid peroxidation in humans (Dietrich-Muszalska et al., 2012).
Although direct evidence linking taxifolin, catechins, and cinnamaldehyde to the inhibition of aggressive behavior is currently limited, their documented neuroprotective and neurotransmitter-modulating effects in various neurodegenerative models provide biochemical support for the observed effects of GDT. Taxifolin has been reviewed to prevent the loss of the dopaminergic nervous system in various neuropathological models, such as Parkinson's, Alzheimer's, and Huntington's diseases, by utilizing anti-inflammatory and antioxidant mechanisms (Yang et al., 2023). Cinnamaldehyde exhibits neuroprotective properties by inhibiting microglial activation and blocking neuroinflammatory pathways, which may ameliorate the pathophysiological environment associated with neuropsychiatric disorders (Hajinejad et al., 2020). Notably, catechins are known to contribute to the maintenance of neurochemical homeostasis in the brain by modulating the activity of GABA and glutamate transporters (Wang et al., 2016). While direct anti-aggressive studies of these individual compounds remain a subject for future research, their collective multi-target neuroprotective mechanisms suggest that they exert a synergistic effect within the GDT formulation to attenuate isolation-induced aggression.
These compounds were predicted to interact with neurotransmission-related targets including dopaminergic, adrenergic, vasopressin, GABAergic, and CRH receptor pathways, all of which play central roles in aggression control and stress adaptation (Haller, 2013; Nelson and Trainor, 2007). Moreover, enrichment analyses highlighted key signaling pathways, such as PI3K-Akt signaling, calcium signaling, neuroactive ligand-receptor interactions, apoptosis regulation, and steroid hormone biosynthesis, which are pathways strongly associated with neuronal survival and plasticity.
CRH receptor networks, adrenergic signaling, and dopaminergic synapse regulation are especially relevant, as dysregulation of these systems is known to underlie isolation-induced hyperarousal and aggressive behavior (Buwalda et al., 2017). The involvement of the PI3K-Akt and calcium signaling pathways parallels previous findings that activation of these cascades supports neurogenesis and ameliorates stress-related neural impairment (Duman and Aghajanian, 2012; Encinas et al., 2006). Taken together, the multi-layered network suggests that GDT exerts its effects not through a single molecular pathway but through coordinated modulation of neuroendocrine-, neurotransmitter-, and neuroplasticity-related systems.
Although GDT significantly improved both behavioral and neurogenic outcomes, DCX expression did not fully recover to sham levels, indicating partial restoration. Additional studies are required to identify the specific active compounds, determine dose-dependent effects, and validate predicted signaling pathways through molecular assays. Moreover, evaluating synaptic markers, such as BDNF, CREB phosphorylation, and inflammatory cytokines, will help to clarify the broader neurobiological effects of GDT.
In summary, behavioral tests, DCX-based neurogenesis evaluation, and network pharmacology converged to show that GDT reduces isolation-induced aggression and promotes hippocampal plasticity through multi-target molecular interactions. These findings highlight GDT as a promising therapeutic candidate for stress-related behavioral dysregulation and underscore the utility of network pharmacology for elucidating the complex actions of traditional medicinal formulations.
Acknowledgments
This study was supported by the Regional Innovation System & Education (RISE) grant through the Gwangju and Jeollanamdo RISE Center (2025-RISE-05-011) and the Jeollanamdo RISE Center (2025-RISE-14-004), funded by the Ministry of Education (MOE), Republic of Korea, as well as by the Convergence Research Group Project (CRC21021) of the National Research Council of Science and Technology.
References
- Ahn GS, Kim SH, Sim BS, Park WS, Song MD, Park JH, Kim DH, Kang JS, Kim JE, Lee SI, Ji GY, Shin SW, Ha GT, Bang JG, Kim JB, Kim TY, Baek SH and Kim YM. (2020). The Interpretation of Shanghanlun(4th ed.). Hani publication. Seoul, Korea. p. 536-539.
-
Alexandrovich Y, Antonov E, Shikhevich S, Kharlamova A, Meister L, Makovka Y, Shepeleva D, Gulevich R and Herbeck Y. (2023). The expression profile of genes associated with behavior, stress, and adult neurogenesis along the hippocampal dorsoventral axis in tame and aggressive foxes. Vavilov Journal of Genetics and Breeding. 27:651.
[https://doi.org/10.18699/VJGB-23-76]

-
Anton A, Schwartz R and Kramer S. (1968). Catecholamines and behavior in isolated and grouped mice. Journal of Psychiatric Research. 6:211-220.
[https://doi.org/10.1016/0022-3956(68)90052-6]

-
Arcusa R, Villaño D, Marhuenda J, Cano M, Cerdà B and Zafrilla P. (2022). Potential role of ginger(Zingiber officinale Roscoe) in the prevention of neurodegenerative diseases. Frontiers in Nutrition. 9:809621.
[https://doi.org/10.3389/fnut.2022.809621]

- Brain P and Nowell N. (1971). Isolation-induced aggression in mice: expectations of social interactions. Communications in Behavioral Biology. 40:203-205.
- Buwalda B, Greerdink M, Vidal J and Koolhaas J. (2017). Social behavior and social stress in adolescence: A neurobiological perspective. Neuroscience & Biobehavioral Reviews. 79:1-12.
-
Cao YJ, Pu ZJ, Tang YP, Shen J, Chen YY, Kang A, Zhou GS and Duan JA. (2017). Advances in bio-active constituents, pharmacology and clinical applications of rhubarb. Chinese Medicine. 12:36.
[https://doi.org/10.1186/s13020-017-0158-5]

-
Chen PY, Wang C, Zhang Y, Yuan C, Yu B, Ke XG, Wu HZ, Yang YF and Xiao XC. (2021). Predicting the molecular mechanism of “Angong Niuhuang Pills” in the treatment of COVID-19 based on network pharmacology. Natural Product Communications. 16:1934578X211024032.
[https://doi.org/10.1177/1934578X211024032]

-
Choi JY, Kang S, Tran MN, Lee S, Ryu SM, Chae SW, Kim DH, Lee YE, Jeong S and Moon C. (2024). Antiepileptic and neuroprotective effects of Rheum tanguticum root extract on trimethyltin-induced epilepsy and neurodegeneration: In Vivo and in Silico analyses. Journal of Integrative Neuroscience. 23:122.
[https://doi.org/10.31083/j.jin2306122]

-
Dietrich-Muszalska A, Kontek B, Olas B and Rabe-Jabłońska J. (2012). Epicatechin inhibits human plasma lipid peroxidation caused by haloperidol in vitro. Neurochemical Research. 37:557-562.
[https://doi.org/10.1007/s11064-011-0642-8]

- Doo IS and Lee SI. (2024). A case report of a patient with insomnia disorder who improved after prescribing Shanghanlun Geijigadaehwang-tang. Journal of Korean Medical Association of Clinical Sanghan-Geumgwe. 16:79-86.
-
Du W, Liang X, Wang S, Lee P and Zhang Y. (2020). The underlying mechanism of Paeonia lactiflora pall. in parkinson’s disease based on a network pharmacology approach. Frontiers in Pharmacology. 11:581984. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2020.581984/full, (cited by 2026 Jan 14).
[https://doi.org/10.3389/fphar.2020.581984]

-
Duman RS and Aghajanian GK. (2012). Synaptic dysfunction in depression: Potential therapeutic targets. Science. 338:68-72.
[https://doi.org/10.1126/science.1222939]

-
Eichelman BS. (1990). Neurochemical and psychopharmacologic aspects of aggressive behavior. Annual Review of Medicine. 41:149-158.
[https://doi.org/10.1146/annurev.me.41.020190.001053]

-
Encinas JM, Vaahtokari A and Enikolopov G. (2006). Fluoxetine targets early progenitor cells in the adult brain. Proceedings of the National Academy of Sciences. 103:8233-8238.
[https://doi.org/10.1073/pnas.0601992103]

-
Fone KC and Porkess MV. (2008). Behavioural and neurochemical effects of post-weaning social isolation in rodents-Relevance to developmental neuropsychiatric disorders. Neuroscience & Biobehavioral Reviews. 32:1087-1102.
[https://doi.org/10.1016/j.neubiorev.2008.03.003]

-
Gfeller D, Grosdidier A, Wirth M, Daina A, Michielin O and Zoete V. (2014). SwissTargetPrediction: A web server for target prediction of bioactive small molecules. Nucleic Acids Research. 42:W32-W38.
[https://doi.org/10.1093/nar/gku293]

-
Guo W, Yao X, Cui R, Yang W and Wang L. (2023). Mechanisms of paeoniaceae action as an antidepressant. Frontiers in Pharmacology. 13:934199. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2022.934199/full, (cited by 2026 Jan 14).
[https://doi.org/10.3389/fphar.2022.934199]

- Ha HY, Yun HJ and Lee SJ. (2020). A study on greater yin disease in Gangpyeong-Shanghanlun by analyzing four mental illness cases. Journal of Korean Medical Association of Clinical Sanghan-Geumgwe. 12:23-50.
-
Hajinejad M, Ghaddaripouri M, Dabzadeh M, Forouzanfar F and Sahab-Negah S. (2020). Natural cinnamaldehyde and its derivatives ameliorate neuroinflammatory pathways in neurodegenerative diseases. BioMed Research International. 2020:1034325.
[https://doi.org/10.1155/2020/1034325]

-
Haller J. (2013). The neurobiology of abnormal manifestations of aggression—a review of hypothalamic mechanisms in cats, rodents, and humans. Brain Research Bulletin. 93:97-109.
[https://doi.org/10.1016/j.brainresbull.2012.10.003]

- Herbology Textbook Committee. (2020). Herbology(4th ed.). Younglim publication. Seoul, Korea. p. 279-281.
-
Hodgins S. (2017). Aggressive behavior among persons with schizophrenia and those who are developing schizophrenia: Attempting to understand the limited evidence on causality. Schizophrenia Bulletin. 43:1021-1026.
[https://doi.org/10.1093/schbul/sbx079]

-
Hu W, Fu W, Wei X, Yang Y, Lu C and Liu Z. (2019). A network pharmacology study on the active ingredients and potential targets of Tripterygium wilfordii hook for treatment of rheumatoid arthritis. Evidence-Based Complementary and Alternative Medicine. 2019:5276865.
[https://doi.org/10.1155/2019/5276865]

-
Huang TT, Leu D and Zou Y. (2015). Oxidative stress and redox regulation on hippocampal-dependent cognitive functions. Archives of Biochemistry and Biophysics. 576:2-7.
[https://doi.org/10.1016/j.abb.2015.03.014]

-
Hwang SH, Choi YG, Jeong MY, Hong YM, Lee JH and Lim S. (2009). Microarray analysis of gene expression profile by treatment of Cinnamomi Ramulus in lipopolysaccharide-stimulated BV-2 cells. Gene. 443:83-90.
[https://doi.org/10.1016/j.gene.2009.04.024]

-
Ibi D, Takuma K, Koike H, Mizoguchi H, Tsuritani K, Kuwahara Y, Kamei H, Nagai T, Yoneda Y and Nabeshima T. (2008). Social isolation rearing-induced impairment of the hippocampal neurogenesis is associated with deficits in spatial memory and emotion-related behaviors in juvenile mice. Journal of Neurochemistry. 105:921-932.
[https://doi.org/10.1111/j.1471-4159.2007.05207.x]

-
Lee AY, Lee JY, and Chun JM. (2020). Exploring the mechanism of Gyejibokryeong-hwan against atherosclerosis using network pharmacology and molecular docking. Plants. 9:1750. https://www.mdpi.com/2223-7747/9/12/1750, (cited by 2026 Jan 14).
[https://doi.org/10.3390/plants9121750]

-
Lee AY, Park W, Kang TW, Cha MH, and Chun JM. (2018). Network pharmacology-based prediction of active compounds and molecular targets in Yijin-Tang acting on hyperlipidaemia and atherosclerosis. Journal of Ethnopharmacology. 221:151-159.
[https://doi.org/10.1016/j.jep.2018.04.027]

-
Lee S, Ryu SM, Kim DH, Lee YE, Lee SJ, Kang S, Kim JS and Lee SI. (2022). Neuroprotective effect of Geijigadaehwang-tang against trimethyltin-induced hippocampal neurodegeneration: An in vitro and in vivo study. Journal of Ethnopharmacology. 296:115451.
[https://doi.org/10.1016/j.jep.2022.115451]

-
Li BJ, Liu P, Chu Z, Shang Y, Huan MX, Dang YH and Gao CG. (2017). Social isolation induces schizophrenia-like behavior potentially associated with HINT1, NMDA receptor 1, and dopamine receptor 2. NeuroReport. 28:462-469.
[https://doi.org/10.1097/WNR.0000000000000775]

-
Li W, Yang Y, Hong L, An FR, Ungvari GS, Ng CH and Xiang YT. (2020). Prevalence of aggression in patients with schizophrenia: A systematic review and meta-analysis of observational studies. Asian Journal of Psychiatry. 47:101846.
[https://doi.org/10.1016/j.ajp.2019.101846]

-
Li X, Chu S, Liu Y and Chen N. (2019). Neuroprotective effects of anthraquinones from rhubarb in central nervous system diseases. Evidence-Based Complementary and Alternative Medicine. 2019:3790728.
[https://doi.org/10.1155/2019/3790728]

-
Li Y and Jiang JG. (2018). Health functions and structure–activity relationships of natural anthraquinones from plants. Food & Function. 9:6063-6080.
[https://doi.org/10.1039/C8FO01569D]

-
Liu X, Fu J, Yao XJ, Yang J, Liu L, Xie TG, Jiang PC, Jiang ZH and Zhu GY. (2018). Phenolic constituents isolated from the twigs of Cinnamomum cassia and their potential neuroprotective effects. Journal of Natural Products. 81:1333-1342.
[https://doi.org/10.1021/acs.jnatprod.7b00924]

-
Miczek KA and O'donnell JM. (1978). Intruder-evoked aggression in isolated and nonisolated mice: effects of psychomotor stimulants and L-dopa. Psychopharmacology. 57:47-55.
[https://doi.org/10.1007/BF00426957]

-
Mitra S, Anjum J, Muni M, Das R, Rauf A, Islam F, Emran TB, Semwal P, Hemeg HA and Alhumaydhi FA. (2022). Exploring the journey of emodin as a potential neuroprotective agent: Novel therapeutic insights with molecular mechanism of action. Biomedicine & Pharmacotherapy. 149:112877.
[https://doi.org/10.1016/j.biopha.2022.112877]

-
Mizuno M, Kawamura H, Ishizuka Y, Sotoyama H and Nawa H. (2010). The anthraquinone derivative emodin attenuates methamphetamine-induced hyperlocomotion and startle response in rats. Pharmacology Biochemistry and Behavior. 97:392-398.
[https://doi.org/10.1016/j.pbb.2010.09.009]

-
Mizuno M, Kawamura H, Takei N and Nawa H. (2008). The anthraquinone derivative Emodin ameliorates neurobehavioral deficits of a rodent model for schizophrenia. Journal of Neural Transmission. 115:521-530.
[https://doi.org/10.1007/s00702-007-0867-5]

-
Mukherjee PK, Banerjee S and Kar A. (2021). Molecular combination networks in medicinal plants: understanding synergy by network pharmacology in Indian traditional medicine. Phytochemistry Reviews. 20:693-703.
[https://doi.org/10.1007/s11101-020-09730-4]

-
Nelson RJ and Trainor BC. (2007). Neural mechanisms of aggression. Nature Reviews Neuroscience. 8:536-546.
[https://doi.org/10.1038/nrn2174]

- Olivier B and Young L. (2002). Animal models of aggression. In Charney D, Davis KL, Coyle JT, et al.(eds.). Neuropsychopharmacology: The Fifth Generation of Progress. Lippincott Williams & Wilkins. Philadelphia, PA, USA. p.1699-1708.
-
Pak SW, Lee AY, Seo YS, Lee SJ, Kim WI, Shin DH, Kim JC, Kim JS, Lim JO and Shin IS. (2022). Anti-asthmatic effects of Phlomis umbrosa Turczaninow using ovalbumin induced asthma murine model and network pharmacology analysis. Biomedicine & Pharmacotherapy. 145:112410.
[https://doi.org/10.1016/j.biopha.2021.112410]

- Park JK. (2020). A Case Report: The effect of Kyejigadahuang-tang on central poststroke pain. Journal of Korean Medical Association of Clinical Sanghan-Geumgwe. 12:125-133.
-
Park SJ, Lee JY, Kim SJ, Choi SY, Yune TY and Ryu JH. (2015). Toll-like receptor-2 deficiency induces schizophrenia-like behaviors in mice. Scientific Reports. 5:8502.
[https://doi.org/10.1038/srep08502]

-
Pastorino G, Cornara L, Soares S, Rodrigues F and Oliveira MBP. (2018). Liquorice(Glycyrrhiza glabra): A phytochemical and pharmacological review. Phytotherapy Research. 32:2323-2339.
[https://doi.org/10.1002/ptr.6178]

-
Qi Y, Zang R, Lu H, Wang Z and Ma Z. (2020). Network pharmacology and bioinformatics approach reveals the hypolipidemic mechanism of Dan Tian Jiang Zhi pill. Medicinal Chemistry Research. 29:243-254.
[https://doi.org/10.1007/s00044-019-02478-3]

-
Ryan SM and Nolan YM. (2016). Neuroinflammation negatively affects adult hippocampal neurogenesis and cognition: Can exercise compensate? Neuroscience & Biobehavioral Reviews. 61:121-131.
[https://doi.org/10.1016/j.neubiorev.2015.12.004]

-
Seibel R, Schneider RH and Gottlieb MG. (2021). Effects of spices(saffron, rosemary, cinnamon, turmeric and ginger) in Alzheimer's disease. Current Alzheimer Research. 18:347-357.
[https://doi.org/10.2174/1567205018666210716122034]

-
Snyder JS, Soumier A, Brewer M, Pickel J and Cameron HA. (2011). Adult hippocampal neurogenesis buffers stress responses and depressive behaviour. Nature. 476:458‐461.
[https://doi.org/10.1038/nature10287]

-
Stranahan AM, Khalil D and Gould E. (2006). Social isolation delays the positive effects of running on adult neurogenesis. Nature Neuroscience. 9:526-533.
[https://doi.org/10.1038/nn1668]

- The Association of Herbal Formula Textbook Publication. (2023). Herbal Formula Science in Korean Medicine: Specifics (Vol. 1) (2nd ed.). Gunja Publishing Co. Paju, Korea. p.29.
-
Toda T, Parylak SL, Linker SB and Gage FH. (2019). The role of adult hippocampal neurogenesis in brain health and disease. Molecular Psychiatry. 24:67-87.
[https://doi.org/10.1038/s41380-018-0036-2]

-
Ulbricht C, Seamon E, Windsor RC, Armbruester N, Bryan JK, Costa D, Giese N, Gruenwald J, Iovin R and Isaac R. (2011). An evidence-based systematic review of cinnamon (Cinnamomum spp.) by the Natural Standard Research Collaboration. Journal of Dietary Supplements. 8:378-454.
[https://doi.org/10.3109/19390211.2011.627783]

-
Upadhyay S, Kumar B and Upadhyay P. (2022). Evaluation of cns depressant and behavioral activity of an ethanol extract of Ziziphus jujuba(ber) in mouse model. Asian Journal of Pharmaceutical and Clinical Research. 15:47-49.
[https://doi.org/10.22159/ajpcr.2022.v15i12.46063]

- Valzelli L. (1969). Aggressive behaviour induced by isolation. In Garattini S and Sigg EB.(eds.). Aggressive Behaviour. Excerpta Medica. Amsterdam, Netherlands. p.70-76.
-
Valzelli L. (1973). The “isolation syndrome” in mice. Psychopharmacologia. 31:305-320.
[https://doi.org/10.1007/BF00421275]

-
Vella D, Marini S, Vitali F, Di Silvestre D, Mauri G and Bellazzi R. (2018). MTGO: PPI network analysis via topological and functional module identification. Scientific Reports. 8:5499.
[https://doi.org/10.1038/s41598-018-23672-0]

-
Wang YX, Engelmann T, Xu YF and Schwarz W. (2016). Catechins from green tea modulate neurotransmitter transporter activity in Xenopus oocytes. Cogent Biology. 2:1261577.
[https://doi.org/10.1080/23312025.2016.1261577]

-
Xu J, Kang F, Wang W, Liu S, Xie J and Yang X. (2022). Comparison between Heat-Clearing Medicine and Antirheumatic Medicine in Treatment of Gastric Cancer Based on Network Pharmacology, Molecular Docking, and Tumor Immune Infiltration Analysis. Evidence-Based Complementary and Alternative Medicine. 2022:7490279.
[https://doi.org/10.1155/2022/7490279]

-
Yang H, Cheng X, Yang YL, Wang YH and Du GH. (2017). Ramulus cinnamomi extract attenuates neuroinflammatory responses via downregulating TLR4/MyD88 signaling pathway in BV2 cells. Neural Regeneration Research. 12:1860-1864.
[https://doi.org/10.4103/1673-5374.219048]

-
Yang R, Yang X and Zhang F. (2023). New perspectives of taxifolin in neurodegenerative diseases. Current Neuropharmacology. 21:2097-2109.
[https://doi.org/10.2174/1570159X21666230203101107]

-
Yavaş SE and Ersoy S. (2025). Effects of different social interactions on hippocampal neurogenesis and neuron numbers in juvenile rats: A histological and behavioral study. Pamukkale Medical Journal. 18:783-793.
[https://doi.org/10.31362/patd.1657975]

- Yen C. (1959). Ataractic supression of isolation induced aggressive behavior. Archives Internationales de Pharmacodynamie et de Therapie. 123:179-185.
- Yun HJ, Ha HY, Doo IS, Lee SI and Lee SJ. (2021). Case series reporting 7 somatic symptom disorder cases diagnosed as greater yin disease by Shanghanlun provisions. Journal of Korean Medical Association of Clinical Sanghan-Geumgwe. 13:45-59.
-
Zhang L, Shi X, Huang Z, Mao J, Mei W, Ding L, Zhang L, Xing R and Wang P. (2020). Network pharmacology approach to uncover the mechanism governing the effect of radix achyranthis bidentatae on osteoarthritis. BMC Complementary Medicine and Therapies. 20:121. https://link.springer.com/article/10.1186/s12906-020-02909-4, (cited by 2026 Jan 14).
[https://doi.org/10.1186/s12906-020-02909-4]